Gregory Stephens, RD, Windrose Partners06.02.14
In 2013 the “Institutional” market for medical nutritional products was about $1.28 billion. Broken down by segment, sales to hospitals were approximately $250 million, nursing homes $540 million and the rapidly growing home care market $490 million. Though the hospital market appears to be smaller, the downstream retail sales for patients after discharge is significant.
The use of dietary supplements in these settings is not new; however, the products administered have typically been multivitamins and minerals used prophylactically (not included in the sales estimates). The medical nutritional segment, often referred to as clinical or therapeutic nutrition, goes beyond these basic supplements. From a regulatory perspective, many products in this category are classified as medical foods.
Medical nutrition products are often formulated to meet the needs of patients unable to consume normal food (e.g., products like Boost and Ensure, which may be fed orally or via nasogastric tube). With advancements in nutritional science we have seen the development of many “disease specific” product formulations.
For example, Abbott Nutrition’s Oxepa is marketed as “Therapeutic Nutrition for Modulating Inflammation.” Through well designed and published clinical trials, Oxepa has been shown to modulate the inflammatory response in critically ill, mechanicallyventilated, tube-fed patients—especially patients with SIRS (systemic inflammatory response syndrome—e.g., sepsis, trauma, burns), ALI (acute lung injury) or ARDS (acute respiratory distress syndrome). Oxepa is calorically dense (1.5 cal/mL) with concentrated calories for fluid-restricted patients, and contains a unique oil blend containing 4.6 g/L of EPA and 4 g/L of GLA to help modulate inflammation.
Disease specific or specialty formulas account for about 40-50% of institutional clinical nutrition sales. Medical devices administering nutritional product sales (e.g., nasogastric tubes, enteral feeding pumps) account for $320 million beyond nutritionals.
Targeting Unmet Needs
The needs of patients and residents differ among the three institutional settings— certainly from the clinicians’ standpoint. Hospitals tend to value disease management (therapeutic) products where the benefits are realized in a relatively short time period. Keep in mind that the average length of stay (ALOS) in hospitals is 4.8 days. A goal is getting the patients well enough to be discharged to go home or stabilized for care in rehabilitation or other long-term care settings. Thus, in hospitals, products that have a disease management or demonstrated cost-reduction impact on patient care in the short-term are more likely to be accepted.
Nursing homes and home care settings, on the other hand, place more value on preventive health benefits. The ALOS in skilled nursing facilities is approximately 2 years (after which 72% expire and 38% are transferred to hospitals or home care). Products that help keep residents healthy and disease free are in demand. For that reason, dietary supplements and products intended to keep healthy individuals well may be more appropriate for these settings.
Strategic Focus
A detailed review of business development strategy elements for the institutional channel is beyond the scope of this column; however, the following insights may be helpful.
Regardless of the clinical setting, focus is critical. There are more than 5,700 hospitals in the U.S. and more than 16,000 skilled nursing facilities. The number of clinical departments, purchase decision makers and influencers makes marketing a daunting task. For perspective, there are more than 1,000 sales representatives covering the clinical nutrition market just for the major medical food companies. Few dietary supplement companies have the resources to provide such coverage. Understanding critical product attributes for success and the decision making process is necessary to assess the market opportunity effectively.
How is this done? A good starting place is to focus on indications with well documented benefits from nutritional intervention. Also, understand the level of clinical substantiation required by decision makers. Clinicians focus more on patient outcomes whereas administration may be more influenced by the cost-benefit of nutritional intervention. Demonstrated product efficacy is of utmost importance in the acute care hospital setting. This goes well beyond the sometimes small, third-party human trials supporting structure-function claims. In this setting, well designed clinical research with appropriate end points are required to support disease management claims and physician expectations.
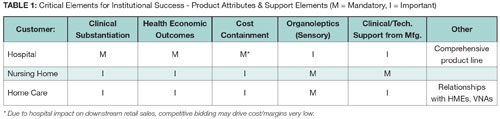
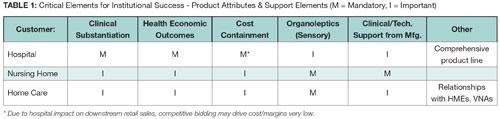
In the nursing home, consider issues like prevention or management of infections or pressure ulcers. A goal of home care is to keep the patient from readmission to the hospital or nursing home and minimize the requirement for more intense, sometimes costly, therapies. In general, maintaining health and wellness carries value. (Select product attributes required for institutional products is summarized in Table 1).
A unique consideration sometimes impacting product pricing to hospitals is the benefit of “downstream” retail sales to discharged patients. Once patients use a product in the hospital and it is recommended they continue consumption after being discharged, then the patient often purchases the product at their local retail establishment, thus increasing sales volume and compliance. Since the ensuing retail sales may be significantly greater than product usage during the relatively short hospital stay, it is important for product samples or coupons to be in the patient’s “Discharge Kit.” This often leads to a competitive “bid” situation between nutritional product companies resulting in declining hospital margins. Regardless, to maximize the potential of ensuing retail sales, hospital staff such as the discharge planner should be included in staff education efforts. Historically, this has been an issue affecting pricing of some meal replacement beverages as well as infant formula sold to hospitals.
The marketing strategy must also reflect and understand decision makers and influencers in the respective institutional settings. The importance of specific health disciplines in adding new products to the formulary, stocking and usage of nutritional products varies. In hospitals, the decision to add a product to the formulary is largely a clinical one, especially for nutritionals targeting new indications. In long-term care settings the decision may be more business related, supported by clinical and cost-benefit research.
Once a product is approved for the formulary, staff education is important to assure it is inventoried and prescribed to the patients. The dietitian and nursing staffs are key influencers in each of the three settings. From a marketing standpoint, to reach the appropriate dietitians, you may choose to target specific “Dietetic Practice Groups” within the Academy of Nutrition and Dietetics (formerly the American Dietetics Association). Practice Groups such as Diabetes Care and Education, Integrative and Functional Medicine, and Healthy Aging and Medical Nutrition Therapy are just a few. Do not overlook other allied health professionals. For patients with diabetes you may consider educating and forming relationships with Certified Diabetes Educators (CDEs), many of which are nurses with a key role in educating patients in blood sugar management. To reach these allied health professionals there are numerous opportunities at conferences, CME sessions and professional associations.
There are many other nuances to the healthcare institutional market for nutritionals. Institutions rely on specialized distributors with their own set of requirements. Companies entering the market may also need to provide resources to address and facilitate the potential of third-party reimbursement.
The market for nutritional products in healthcare institutions is large and broadening from the traditional meal replacement beverages. Supplements like omega-3s and probiotics are making inroads, and more products will follow as clinical documentation of efficacy increases. This market is well developed and the decision makers are more sophisticated from a clinical perspective than traditional dietary supplement distribution channels. A company interested in investigating institutional distribution would be wise to seek counseling from a qualified consultant or other persons who have experience and have successfully launched products in this expanding new market.
Greg Stephens, RD, is president of Windrose Partners, a company serving clients in the the dietary supplement, functional food and natural product industries. Formerly vice president of strategic consulting with the Natural Marketing Institute (NMI) and vice president of sales and marketing for Nurture, Inc (OatVantage), he has 25 years of specialized expertise in the nutritional and pharmaceutical industries. His prior experience includes a progressive series of senior management positions with Abbott Nutrition (Ross Products Division of Abbott Laboratories), including development of global nutrition strategies for disease-specific growth platforms and business development for Abbott’s medical foods portfolio. He can be reached at 215-860-5186; E-mail: gregstephens@windrosepartners.com.
The use of dietary supplements in these settings is not new; however, the products administered have typically been multivitamins and minerals used prophylactically (not included in the sales estimates). The medical nutritional segment, often referred to as clinical or therapeutic nutrition, goes beyond these basic supplements. From a regulatory perspective, many products in this category are classified as medical foods.
Medical nutrition products are often formulated to meet the needs of patients unable to consume normal food (e.g., products like Boost and Ensure, which may be fed orally or via nasogastric tube). With advancements in nutritional science we have seen the development of many “disease specific” product formulations.
For example, Abbott Nutrition’s Oxepa is marketed as “Therapeutic Nutrition for Modulating Inflammation.” Through well designed and published clinical trials, Oxepa has been shown to modulate the inflammatory response in critically ill, mechanicallyventilated, tube-fed patients—especially patients with SIRS (systemic inflammatory response syndrome—e.g., sepsis, trauma, burns), ALI (acute lung injury) or ARDS (acute respiratory distress syndrome). Oxepa is calorically dense (1.5 cal/mL) with concentrated calories for fluid-restricted patients, and contains a unique oil blend containing 4.6 g/L of EPA and 4 g/L of GLA to help modulate inflammation.
Disease specific or specialty formulas account for about 40-50% of institutional clinical nutrition sales. Medical devices administering nutritional product sales (e.g., nasogastric tubes, enteral feeding pumps) account for $320 million beyond nutritionals.
Targeting Unmet Needs
The needs of patients and residents differ among the three institutional settings— certainly from the clinicians’ standpoint. Hospitals tend to value disease management (therapeutic) products where the benefits are realized in a relatively short time period. Keep in mind that the average length of stay (ALOS) in hospitals is 4.8 days. A goal is getting the patients well enough to be discharged to go home or stabilized for care in rehabilitation or other long-term care settings. Thus, in hospitals, products that have a disease management or demonstrated cost-reduction impact on patient care in the short-term are more likely to be accepted.
Nursing homes and home care settings, on the other hand, place more value on preventive health benefits. The ALOS in skilled nursing facilities is approximately 2 years (after which 72% expire and 38% are transferred to hospitals or home care). Products that help keep residents healthy and disease free are in demand. For that reason, dietary supplements and products intended to keep healthy individuals well may be more appropriate for these settings.
Strategic Focus
A detailed review of business development strategy elements for the institutional channel is beyond the scope of this column; however, the following insights may be helpful.
Regardless of the clinical setting, focus is critical. There are more than 5,700 hospitals in the U.S. and more than 16,000 skilled nursing facilities. The number of clinical departments, purchase decision makers and influencers makes marketing a daunting task. For perspective, there are more than 1,000 sales representatives covering the clinical nutrition market just for the major medical food companies. Few dietary supplement companies have the resources to provide such coverage. Understanding critical product attributes for success and the decision making process is necessary to assess the market opportunity effectively.
How is this done? A good starting place is to focus on indications with well documented benefits from nutritional intervention. Also, understand the level of clinical substantiation required by decision makers. Clinicians focus more on patient outcomes whereas administration may be more influenced by the cost-benefit of nutritional intervention. Demonstrated product efficacy is of utmost importance in the acute care hospital setting. This goes well beyond the sometimes small, third-party human trials supporting structure-function claims. In this setting, well designed clinical research with appropriate end points are required to support disease management claims and physician expectations.
In the nursing home, consider issues like prevention or management of infections or pressure ulcers. A goal of home care is to keep the patient from readmission to the hospital or nursing home and minimize the requirement for more intense, sometimes costly, therapies. In general, maintaining health and wellness carries value. (Select product attributes required for institutional products is summarized in Table 1).
A unique consideration sometimes impacting product pricing to hospitals is the benefit of “downstream” retail sales to discharged patients. Once patients use a product in the hospital and it is recommended they continue consumption after being discharged, then the patient often purchases the product at their local retail establishment, thus increasing sales volume and compliance. Since the ensuing retail sales may be significantly greater than product usage during the relatively short hospital stay, it is important for product samples or coupons to be in the patient’s “Discharge Kit.” This often leads to a competitive “bid” situation between nutritional product companies resulting in declining hospital margins. Regardless, to maximize the potential of ensuing retail sales, hospital staff such as the discharge planner should be included in staff education efforts. Historically, this has been an issue affecting pricing of some meal replacement beverages as well as infant formula sold to hospitals.
The marketing strategy must also reflect and understand decision makers and influencers in the respective institutional settings. The importance of specific health disciplines in adding new products to the formulary, stocking and usage of nutritional products varies. In hospitals, the decision to add a product to the formulary is largely a clinical one, especially for nutritionals targeting new indications. In long-term care settings the decision may be more business related, supported by clinical and cost-benefit research.
Once a product is approved for the formulary, staff education is important to assure it is inventoried and prescribed to the patients. The dietitian and nursing staffs are key influencers in each of the three settings. From a marketing standpoint, to reach the appropriate dietitians, you may choose to target specific “Dietetic Practice Groups” within the Academy of Nutrition and Dietetics (formerly the American Dietetics Association). Practice Groups such as Diabetes Care and Education, Integrative and Functional Medicine, and Healthy Aging and Medical Nutrition Therapy are just a few. Do not overlook other allied health professionals. For patients with diabetes you may consider educating and forming relationships with Certified Diabetes Educators (CDEs), many of which are nurses with a key role in educating patients in blood sugar management. To reach these allied health professionals there are numerous opportunities at conferences, CME sessions and professional associations.
There are many other nuances to the healthcare institutional market for nutritionals. Institutions rely on specialized distributors with their own set of requirements. Companies entering the market may also need to provide resources to address and facilitate the potential of third-party reimbursement.
The market for nutritional products in healthcare institutions is large and broadening from the traditional meal replacement beverages. Supplements like omega-3s and probiotics are making inroads, and more products will follow as clinical documentation of efficacy increases. This market is well developed and the decision makers are more sophisticated from a clinical perspective than traditional dietary supplement distribution channels. A company interested in investigating institutional distribution would be wise to seek counseling from a qualified consultant or other persons who have experience and have successfully launched products in this expanding new market.
Greg Stephens, RD, is president of Windrose Partners, a company serving clients in the the dietary supplement, functional food and natural product industries. Formerly vice president of strategic consulting with the Natural Marketing Institute (NMI) and vice president of sales and marketing for Nurture, Inc (OatVantage), he has 25 years of specialized expertise in the nutritional and pharmaceutical industries. His prior experience includes a progressive series of senior management positions with Abbott Nutrition (Ross Products Division of Abbott Laboratories), including development of global nutrition strategies for disease-specific growth platforms and business development for Abbott’s medical foods portfolio. He can be reached at 215-860-5186; E-mail: gregstephens@windrosepartners.com.