Shyam Gupta, PhD, Bioderm Research09.01.14
The popularity of being beautiful inside and out proves the old adage, “cosmeceuticals are for topicals and nutraceuticals are for ingestibles,” is a myth. The use of cosmeceutical ingredients in nutraceutical applications has become quite prevalent; the reverse, however, is in its nascent stage. That’s because topical delivery of natural cosmeceutical and nutraceutical ingredients presents challenges unique to the chemistry of specific ingredients and the mode of their desired biological function.
Nutritional supplements are consumed by direct ingestion. The ingestion method can pose certain issues relative to their unacceptable odor and taste, and degradation of beneficial active agents during the body’s digestive and absorptive processes. Topically delivered formulations circumvent some of these concerns due to their application near or at the site of the skin condition or affliction.
Beauty inside & out is quickly becoming a new trend in marketing strategies to provide consumer satisfaction overall. This article presents combinatorial inside and outside formulation technologies utilizing popular cosmeceutical and nutraceutical ingredients in skin clarifying and anti-aging applications.
Formulation Challenges
Consumers seldom notice the physical state of the functional ingredients in a nutritional supplement due to the product’s delivery system (for example, a capsule or a tablet with an outer coating to mask any off-odor, or a sweetened syrup to mask its bitter taste). However, the appearance and aroma of a topical product plays a pivotal role in the consumer psychology; a cream or lotion that is pure white in appearance with a creamy, silky-smooth tactile feel is preferred globally. This can limit the utilization of some of the most effective ingredients, for example certain antioxidants, in a topical cream or lotion in any beneficial amount.
At just 0.1%, astaxanthin, a bright red carotenoid with potent antioxidant attributes, can render a yellow-to-orange tint to a topical cream or lotion formulation, and even stain clothing. Curcumin from turmeric is a potent antioxidant with a bright yellow-to-orange color that can leave a yellow color on skin when applied topically. Similarly, the juice from highly colored fruits and berries (such as grape, blueberry, mango, orange, strawberry and blackberry), which are healthful nutritional agents, cannot be used directly in cosmetic formulations due to consumer aesthetics and formulation stability concerns.
These issues can be resolved by the isolation of purified active agents from their natural states, or by their chemical modification. The hydrogenation of curcumin provides almost-white tetrahydrocurcuminoids that are potent antioxidants without the consumer concerns of curcumin’s color. However, such chemical modifications are not always successful in providing ingredients that maintain their desirable efficacy. This dilemma of inherent problems with the direct use of ingredients in their natural, unaltered state in topical preparations provides an opportunity for a dual inside-outside marketing strategy.
Quorum-Sensing Ginger
Rhizomes of the ginger family contain some of the most well known anti-arthritis and anti-inflammatory ingredients known to pharmacy. Ginger has been used in Ayurvedic and Tibetan medicine for centuries (oral, or “inside” application). Ginger contains essential oils and spicy substances, such as gingerol, shogaol, zingerone and capsaicin, that are principally responsible for its curative properties. Recent research has paved the way for new topical (“outside”) applications for ginger.
Quorum sensing is a system that regulates gene expression in bacteria that has become a target for the development of (anti-quorum sensing) novel anti-infective treatments that do not rely on the use of antibiotics.1 Anti-quorum sensing has been a promising strategy to combat bacterial infections, as it is unlikely to develop drug-resistant pathogens. A number of anti-quorum sensing plant-based natural products and their anti-quorum sensing mechanisms have been extensively studied in this context.
Phenolic components of ginger (Zingiber officinale), for example gingerol, shogaol and zingerone, have exhibited quorum-sensing inhibitory activity (QSI) against Chromobacterium violaceum and Pseudomonas aeruginosa.2 Chemical modification of shogaol to azashogaol and an isoxazoline derivative of gingerol were prepared, both of which exhibited good QSI activity. This paves the way for the development of new anti-infective topical applications for purified ginger extracts that contain gingerol, shogaol and/or zingerone and their chemical derivatives. Inhibition of hair growth and skin cancer by gingerol is noteworthy.3
On the oral side, ginger is effective in alleviating chemotherapy-induced nausea and emesis. Ginger-mediated antiemetic effect has been attributed to its inhibition of serotonin (5-HT) receptor activity. Oral administration of gingerol, shogaol and/or zingerone, which inhibited 5-HT responses in a dose dependent manner, has been shown to reduce emesis in cancer patients.4 Ginger has been used to treat post-operative nausea and vomiting in a recent clinical trial.5 Oral intake of ginger powder has shown clinical efficacy in the treatment of insulin resistance.6
6-Dehydrogingerdione (6-DG), a chemical derivative of Zingiber officinale, is a recent introduction for human skin wound repair. 6-DG treatment accelerated cellular proliferation and migration dose-dependently. Enzyme-linked immunosorbent assay showed that 6-DG brought about higher growth factor productions. 6-DG increased fibroblast collagen, reduced matrix metalloproteinase-1 (MMP-1) protein expression and recovered tissue inhibitor of metalloproteinase-1 (TIMP-1) secretion. 6-DG, therefore, has the potential to be a novel agent for human skin repair.7 Another relative of ginger, Kaempferia galanga, has exhibited exciting new applications including protection from skin photoaging.
Ginger ingredients offer dual excitement (via oral and topical delivery systems) in the development of new consumer products.
Mango, sometimes called “The King of Fruits,” is a highly prized fruit in Asia and Central and South America. Mango deserves better recognition in oral and topical formulations for total body care. From its leaves to fruit pulp to fruit skin to seed, mango is blessed with cosmeceutical agents. Mango peel and kernel offer one of the most underutilized sources of cosmeceuticals, yet several million pounds of mango seed worldwide are either burned or allowed to rot each year. Mango seed oil and mango butter are one of the best exotic natural fat-based cosmeceuticals for skin repair.9 Extract from mango leaves has anti-tyrosinase (skin whitening), anti-inflammatory and antioxidant activity.10
Mangiferin, a compound obtained from mango, inhibits ubiquitination, provides antioxidant and cytoprotective action, protection against radiation-induced DNA damage and UVB-induced skin aging.11 Mangiferin has also been shown to inhibit matrix metalloproteinase-1 induced by oxidative stress and inflammation. Mangiferin was found to scavenge intracellular reactive oxygen species (ROS), superoxide radicals and hydroxyl radicals using human skin keratinocytes, thus directing its application in skin anti-aging formulations; albeit with some caution directed to its light yellow color.12 Cellulose nanocrystals, recently obtained from mango, have potential as a delivery system for cosmetic and pharmaceutical agents.13 Mango bark extract needs commercialization in skin care formulations for its potent antioxidant properties.14
The bioactive compounds in mango peel extracts recently identified by HPLC were protocatechuic acid, gentisic acid and gallic acid, syringic acid, mangiferin, ellagic acid, gentisyl-protocatechuic acid and quercetin in both raw and ripe peels. The raw peel also showed the presence of glycosylated iriflophenone and maclurin derivatives. Beta-carotene was the major carotenoid followed by violaxanthin and lutein.15 Ingredient suppliers should consider the development of both iriflophenone and maclurin for anticipated antioxidant, anti-inflammatory, MMP-inhibiting, skin brightening, UV-absorbing and skin anti-aging applications.
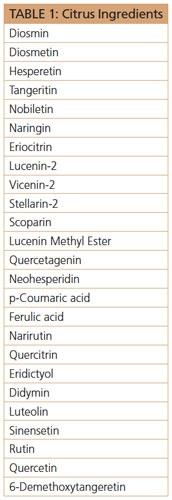
Comparing apples to oranges may be a laughable subject in social circles. However, utilizing ingredients from apples and oranges can provide unprecedented marketing opportunities in oral and topical formulations in view of recent research. Oranges, for example, contain a surprising array of polyphenolic compounds (see Table 1) with attributes that include potent antioxidant, anti-inflammatory, tyrosinase-inhibiting, MMP-inhibiting, collagen-building, antiglycation and activation of mitochondrial biogenesis.16
Citrus press-cakes, by-products of the juice industry, have found application as whitening agents in the cosmetic industry via their down-regulation of tyrosinase, TRP-1, TRP-2 and MITF expressions.17 Clearly, a number of these ingredients offer opportunities for inside (nutritional) and outside (topical) marketing innovations.
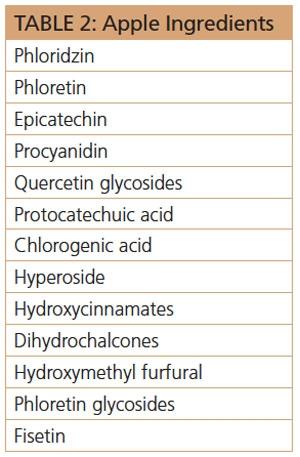
Apples contain some of the best known ingredients (see Table 2) that possess antioxidant, anti-inflammatory, wrinkle reduction, skin brightening, and skin anti-aging benefits.18 Two of the most noteworthy ingredients from apple, phloridzin and phloretin, are obtained from apple root. These two potent antioxidant, anti-inflammatories offer these benefits: MMP-inhibition, free-radical scavenging, superoxide dismutase (SOD) activation, glutathione peroxidase activation, peroxynitrite scavenging, lipid peroxidation inhibition, and, as if that weren’t enough, lipoprotein oxidation inhibition, suppression of stimulated expression of endothelial adhesion molecules and deactivation of human platelets.19
Nutritionally speaking, the phrase, “An apple a day helps keep the doctor away,” most probably relates to the aforementioned attributes, as consumption of apple fruits along with peel might be recommended to gain better nutritive benefits. To add more, an apple a day may help keep young women in a better state of their sexual life.20
From skin to seed and pulp in between, apple provides a plethora of phyto- and micro-nutrients. Apple seed oil, obtained from apple pomace, is comparable with edible food oil in its physical properties, indicating its better stability and broad application in the food, cosmetic and pharmaceutical industries. Apple seed oil is also a good source of natural antioxidants.21 Apple wax, obtained from apple skin, is highly prized due to its limited availability. It provides an unusually high gloss and shine in hair and nail care cosmetic products. “Apple Polishing” may have brought its brilliant results from the shine of this wax!
Grape Ingredients
Resveratrol and grape seed oil are two of the most promoted ingredients currently utilized in nutritional and cosmetic applications. Resveratrol is only one of more than 20 known hydroxystilbenes (in addition to its dimers and hexamers) isolated from Vitis vinifera. Grapes, from seed to pulp to skin, are infused with additional exciting phytochemicals that deserve a spotlight in nutritional and cosmetic formulations, examples of which can be seen in Table 3 (some duplicated with possible alternate names).22 Vam-3 (trans-epsilon-viniferin), a resveratrol dimer, inhibits cigarette smoke-induced cell apoptosis in lungs by improving mitochondrial function.23 This enhancement of mitochondrial function could play a role in skin anti-aging, too.
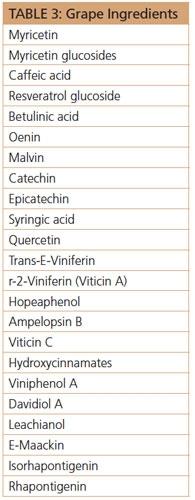
Recently-discovered resveratrol oligomers, trans-epsilon-viniferin and r-2-viniferin, are inhibitors of cystic fibrosis transmembrane conductance regulators.24 Unusual chemical structure of these oligomers, for example trans-epsilon-viniferin, would suggest application in anti-aging and skin brightening products. Betulinic acid, isolated from Vitis amurensis root, attenuates melanin production by inhibiting tyrosinase, tyrosinase-related protein (TRP)-1 and TRP-2 expression through the modulation of their corresponding transcription factors.25 This paves the way for application in skin whitening and age-spot reduction formulations. It is quite surprising that betulinic acid does not possess a dihydroxybenzene moiety, which is so prevalent in other tyrosinase inhibitors; for example, resveratrol has shown potent inhibitory effect on melanin synthesis via reduction in tyrosinase-related protein 2 among melanogenic enzymes. This is the first evidence supporting resveratrol as a depigmentation agent, and its application in treating hyperpigmentation and skin photoaging.26
Formulation Strategies
In formulating oral treatments, the delivery systems are rather straightforward: a liquid, pill, powder, capsule or paste that is quickly swallowed. Consumers generally do not concern themselves with the color, odor or physical appearance of such formulations.
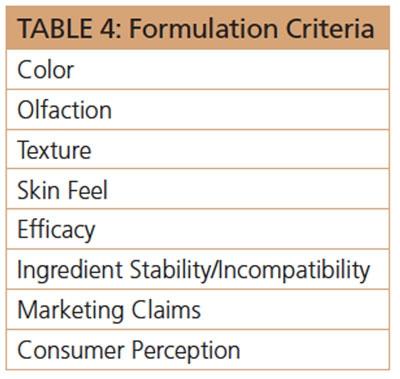
However, for topical preparations, all of the above become of paramount importance. A skin lotion that can discolor skin or clothing, an anti-aging cream that has an off-odor or a gel that has separated into layers all render a product unacceptable to the consumer. The proper selection of ingredients in a formulation thus becomes a major issue. It is preferred that the ingredients are white in color, or as light as possible, so they do not impart a perceptible color to the final formulation.
To illustrate this point, all phenolic and polyphenolic antioxidants discolor when exposed to air and/or light. Special handling is required in the manufacture and packaging of formulations that contain such ingredients. The odor of the formulation, before any fragrance is added, should be neutral with no hint of a “chemical odor,” which signals harsh, synthetic materials to the consumer. The formula should have a smooth texture as well. A lotion must flow uniformly and not be gloppy. A cream should spread easily and evenly upon application. The formula should provide a silky smooth and non-oily powdery skin feel; and it should contain ingredients that signal “immediate” efficacy.
For example, inclusion of a small amount of titanium dioxide in a skin whitening formula, along with functional ingredients that may require some time to show their efficacy, provides the consumer a signal that skin is becoming of a lighter tone even after just one application. Of course, the product claims may state that the skin whitening benefits are to become perceptible after two or more weeks of use. This exemplifies the discord between the consumer perception versus the consumer expectation. The formula must have ingredient compatibility, and provide shelf-stability for at least a two-year time period.
Yet, even after all of this has been achieved and backed with careful product testing, both in the lab and by the consumer, a great formula in an unattractive or non-functional packaging can trigger product failure in the marketplace. The area of consumerism that relates to complex and unpredictable psychological issues of expectation versus perception of a product’s efficacy, which is further influenced by marketing claims and advertising promotion, remains more of a mystique than a reliable science or art. For example, currently popular “back to nature” consumer preference has cast a shadow on certain synthetic ingredients that, otherwise, have a solid track record of safety and efficacy. The use of such “chemicals” in a formula, despite their desirable performance and consumer appeal, can prove fatal for that product in the marketplace.
Marketers and formulators of cosmetic products have an insatiable quest for new, high performance nature-based ingredients. This article draws to the attention of ingredient suppliers, formulators and marketers innovative ingredients from the same age-old and time-tested popular natural sources that await their commercialization in both nutritional and topical consumer products for an inside-outside strategy to benefit the consumer.
Shyam Gupta is an international consultant in innovative skin and hair care ingredients and delivery systems with more than 100 patents, patent applications, cosmetic publications and book chapters specializing in natural and science-based formulations with enhanced efficacy and consumer desirable performance attributes. Phone: 602-996-9700; E-mail: shyam@biodermresearch.com;
Website: www.biodermresearch.com
References
Nutritional supplements are consumed by direct ingestion. The ingestion method can pose certain issues relative to their unacceptable odor and taste, and degradation of beneficial active agents during the body’s digestive and absorptive processes. Topically delivered formulations circumvent some of these concerns due to their application near or at the site of the skin condition or affliction.
Beauty inside & out is quickly becoming a new trend in marketing strategies to provide consumer satisfaction overall. This article presents combinatorial inside and outside formulation technologies utilizing popular cosmeceutical and nutraceutical ingredients in skin clarifying and anti-aging applications.
Formulation Challenges
Consumers seldom notice the physical state of the functional ingredients in a nutritional supplement due to the product’s delivery system (for example, a capsule or a tablet with an outer coating to mask any off-odor, or a sweetened syrup to mask its bitter taste). However, the appearance and aroma of a topical product plays a pivotal role in the consumer psychology; a cream or lotion that is pure white in appearance with a creamy, silky-smooth tactile feel is preferred globally. This can limit the utilization of some of the most effective ingredients, for example certain antioxidants, in a topical cream or lotion in any beneficial amount.
At just 0.1%, astaxanthin, a bright red carotenoid with potent antioxidant attributes, can render a yellow-to-orange tint to a topical cream or lotion formulation, and even stain clothing. Curcumin from turmeric is a potent antioxidant with a bright yellow-to-orange color that can leave a yellow color on skin when applied topically. Similarly, the juice from highly colored fruits and berries (such as grape, blueberry, mango, orange, strawberry and blackberry), which are healthful nutritional agents, cannot be used directly in cosmetic formulations due to consumer aesthetics and formulation stability concerns.
These issues can be resolved by the isolation of purified active agents from their natural states, or by their chemical modification. The hydrogenation of curcumin provides almost-white tetrahydrocurcuminoids that are potent antioxidants without the consumer concerns of curcumin’s color. However, such chemical modifications are not always successful in providing ingredients that maintain their desirable efficacy. This dilemma of inherent problems with the direct use of ingredients in their natural, unaltered state in topical preparations provides an opportunity for a dual inside-outside marketing strategy.
Quorum-Sensing Ginger
Rhizomes of the ginger family contain some of the most well known anti-arthritis and anti-inflammatory ingredients known to pharmacy. Ginger has been used in Ayurvedic and Tibetan medicine for centuries (oral, or “inside” application). Ginger contains essential oils and spicy substances, such as gingerol, shogaol, zingerone and capsaicin, that are principally responsible for its curative properties. Recent research has paved the way for new topical (“outside”) applications for ginger.
Quorum sensing is a system that regulates gene expression in bacteria that has become a target for the development of (anti-quorum sensing) novel anti-infective treatments that do not rely on the use of antibiotics.1 Anti-quorum sensing has been a promising strategy to combat bacterial infections, as it is unlikely to develop drug-resistant pathogens. A number of anti-quorum sensing plant-based natural products and their anti-quorum sensing mechanisms have been extensively studied in this context.
Phenolic components of ginger (Zingiber officinale), for example gingerol, shogaol and zingerone, have exhibited quorum-sensing inhibitory activity (QSI) against Chromobacterium violaceum and Pseudomonas aeruginosa.2 Chemical modification of shogaol to azashogaol and an isoxazoline derivative of gingerol were prepared, both of which exhibited good QSI activity. This paves the way for the development of new anti-infective topical applications for purified ginger extracts that contain gingerol, shogaol and/or zingerone and their chemical derivatives. Inhibition of hair growth and skin cancer by gingerol is noteworthy.3
On the oral side, ginger is effective in alleviating chemotherapy-induced nausea and emesis. Ginger-mediated antiemetic effect has been attributed to its inhibition of serotonin (5-HT) receptor activity. Oral administration of gingerol, shogaol and/or zingerone, which inhibited 5-HT responses in a dose dependent manner, has been shown to reduce emesis in cancer patients.4 Ginger has been used to treat post-operative nausea and vomiting in a recent clinical trial.5 Oral intake of ginger powder has shown clinical efficacy in the treatment of insulin resistance.6
6-Dehydrogingerdione (6-DG), a chemical derivative of Zingiber officinale, is a recent introduction for human skin wound repair. 6-DG treatment accelerated cellular proliferation and migration dose-dependently. Enzyme-linked immunosorbent assay showed that 6-DG brought about higher growth factor productions. 6-DG increased fibroblast collagen, reduced matrix metalloproteinase-1 (MMP-1) protein expression and recovered tissue inhibitor of metalloproteinase-1 (TIMP-1) secretion. 6-DG, therefore, has the potential to be a novel agent for human skin repair.7 Another relative of ginger, Kaempferia galanga, has exhibited exciting new applications including protection from skin photoaging.
Ginger ingredients offer dual excitement (via oral and topical delivery systems) in the development of new consumer products.
Mango, sometimes called “The King of Fruits,” is a highly prized fruit in Asia and Central and South America. Mango deserves better recognition in oral and topical formulations for total body care. From its leaves to fruit pulp to fruit skin to seed, mango is blessed with cosmeceutical agents. Mango peel and kernel offer one of the most underutilized sources of cosmeceuticals, yet several million pounds of mango seed worldwide are either burned or allowed to rot each year. Mango seed oil and mango butter are one of the best exotic natural fat-based cosmeceuticals for skin repair.9 Extract from mango leaves has anti-tyrosinase (skin whitening), anti-inflammatory and antioxidant activity.10
Mangiferin, a compound obtained from mango, inhibits ubiquitination, provides antioxidant and cytoprotective action, protection against radiation-induced DNA damage and UVB-induced skin aging.11 Mangiferin has also been shown to inhibit matrix metalloproteinase-1 induced by oxidative stress and inflammation. Mangiferin was found to scavenge intracellular reactive oxygen species (ROS), superoxide radicals and hydroxyl radicals using human skin keratinocytes, thus directing its application in skin anti-aging formulations; albeit with some caution directed to its light yellow color.12 Cellulose nanocrystals, recently obtained from mango, have potential as a delivery system for cosmetic and pharmaceutical agents.13 Mango bark extract needs commercialization in skin care formulations for its potent antioxidant properties.14
The bioactive compounds in mango peel extracts recently identified by HPLC were protocatechuic acid, gentisic acid and gallic acid, syringic acid, mangiferin, ellagic acid, gentisyl-protocatechuic acid and quercetin in both raw and ripe peels. The raw peel also showed the presence of glycosylated iriflophenone and maclurin derivatives. Beta-carotene was the major carotenoid followed by violaxanthin and lutein.15 Ingredient suppliers should consider the development of both iriflophenone and maclurin for anticipated antioxidant, anti-inflammatory, MMP-inhibiting, skin brightening, UV-absorbing and skin anti-aging applications.
Comparing apples to oranges may be a laughable subject in social circles. However, utilizing ingredients from apples and oranges can provide unprecedented marketing opportunities in oral and topical formulations in view of recent research. Oranges, for example, contain a surprising array of polyphenolic compounds (see Table 1) with attributes that include potent antioxidant, anti-inflammatory, tyrosinase-inhibiting, MMP-inhibiting, collagen-building, antiglycation and activation of mitochondrial biogenesis.16
Citrus press-cakes, by-products of the juice industry, have found application as whitening agents in the cosmetic industry via their down-regulation of tyrosinase, TRP-1, TRP-2 and MITF expressions.17 Clearly, a number of these ingredients offer opportunities for inside (nutritional) and outside (topical) marketing innovations.
Apples contain some of the best known ingredients (see Table 2) that possess antioxidant, anti-inflammatory, wrinkle reduction, skin brightening, and skin anti-aging benefits.18 Two of the most noteworthy ingredients from apple, phloridzin and phloretin, are obtained from apple root. These two potent antioxidant, anti-inflammatories offer these benefits: MMP-inhibition, free-radical scavenging, superoxide dismutase (SOD) activation, glutathione peroxidase activation, peroxynitrite scavenging, lipid peroxidation inhibition, and, as if that weren’t enough, lipoprotein oxidation inhibition, suppression of stimulated expression of endothelial adhesion molecules and deactivation of human platelets.19
Nutritionally speaking, the phrase, “An apple a day helps keep the doctor away,” most probably relates to the aforementioned attributes, as consumption of apple fruits along with peel might be recommended to gain better nutritive benefits. To add more, an apple a day may help keep young women in a better state of their sexual life.20
From skin to seed and pulp in between, apple provides a plethora of phyto- and micro-nutrients. Apple seed oil, obtained from apple pomace, is comparable with edible food oil in its physical properties, indicating its better stability and broad application in the food, cosmetic and pharmaceutical industries. Apple seed oil is also a good source of natural antioxidants.21 Apple wax, obtained from apple skin, is highly prized due to its limited availability. It provides an unusually high gloss and shine in hair and nail care cosmetic products. “Apple Polishing” may have brought its brilliant results from the shine of this wax!
Grape Ingredients
Resveratrol and grape seed oil are two of the most promoted ingredients currently utilized in nutritional and cosmetic applications. Resveratrol is only one of more than 20 known hydroxystilbenes (in addition to its dimers and hexamers) isolated from Vitis vinifera. Grapes, from seed to pulp to skin, are infused with additional exciting phytochemicals that deserve a spotlight in nutritional and cosmetic formulations, examples of which can be seen in Table 3 (some duplicated with possible alternate names).22 Vam-3 (trans-epsilon-viniferin), a resveratrol dimer, inhibits cigarette smoke-induced cell apoptosis in lungs by improving mitochondrial function.23 This enhancement of mitochondrial function could play a role in skin anti-aging, too.
Recently-discovered resveratrol oligomers, trans-epsilon-viniferin and r-2-viniferin, are inhibitors of cystic fibrosis transmembrane conductance regulators.24 Unusual chemical structure of these oligomers, for example trans-epsilon-viniferin, would suggest application in anti-aging and skin brightening products. Betulinic acid, isolated from Vitis amurensis root, attenuates melanin production by inhibiting tyrosinase, tyrosinase-related protein (TRP)-1 and TRP-2 expression through the modulation of their corresponding transcription factors.25 This paves the way for application in skin whitening and age-spot reduction formulations. It is quite surprising that betulinic acid does not possess a dihydroxybenzene moiety, which is so prevalent in other tyrosinase inhibitors; for example, resveratrol has shown potent inhibitory effect on melanin synthesis via reduction in tyrosinase-related protein 2 among melanogenic enzymes. This is the first evidence supporting resveratrol as a depigmentation agent, and its application in treating hyperpigmentation and skin photoaging.26
Formulation Strategies
In formulating oral treatments, the delivery systems are rather straightforward: a liquid, pill, powder, capsule or paste that is quickly swallowed. Consumers generally do not concern themselves with the color, odor or physical appearance of such formulations.
However, for topical preparations, all of the above become of paramount importance. A skin lotion that can discolor skin or clothing, an anti-aging cream that has an off-odor or a gel that has separated into layers all render a product unacceptable to the consumer. The proper selection of ingredients in a formulation thus becomes a major issue. It is preferred that the ingredients are white in color, or as light as possible, so they do not impart a perceptible color to the final formulation.
To illustrate this point, all phenolic and polyphenolic antioxidants discolor when exposed to air and/or light. Special handling is required in the manufacture and packaging of formulations that contain such ingredients. The odor of the formulation, before any fragrance is added, should be neutral with no hint of a “chemical odor,” which signals harsh, synthetic materials to the consumer. The formula should have a smooth texture as well. A lotion must flow uniformly and not be gloppy. A cream should spread easily and evenly upon application. The formula should provide a silky smooth and non-oily powdery skin feel; and it should contain ingredients that signal “immediate” efficacy.
For example, inclusion of a small amount of titanium dioxide in a skin whitening formula, along with functional ingredients that may require some time to show their efficacy, provides the consumer a signal that skin is becoming of a lighter tone even after just one application. Of course, the product claims may state that the skin whitening benefits are to become perceptible after two or more weeks of use. This exemplifies the discord between the consumer perception versus the consumer expectation. The formula must have ingredient compatibility, and provide shelf-stability for at least a two-year time period.
Yet, even after all of this has been achieved and backed with careful product testing, both in the lab and by the consumer, a great formula in an unattractive or non-functional packaging can trigger product failure in the marketplace. The area of consumerism that relates to complex and unpredictable psychological issues of expectation versus perception of a product’s efficacy, which is further influenced by marketing claims and advertising promotion, remains more of a mystique than a reliable science or art. For example, currently popular “back to nature” consumer preference has cast a shadow on certain synthetic ingredients that, otherwise, have a solid track record of safety and efficacy. The use of such “chemicals” in a formula, despite their desirable performance and consumer appeal, can prove fatal for that product in the marketplace.
Marketers and formulators of cosmetic products have an insatiable quest for new, high performance nature-based ingredients. This article draws to the attention of ingredient suppliers, formulators and marketers innovative ingredients from the same age-old and time-tested popular natural sources that await their commercialization in both nutritional and topical consumer products for an inside-outside strategy to benefit the consumer.
Shyam Gupta is an international consultant in innovative skin and hair care ingredients and delivery systems with more than 100 patents, patent applications, cosmetic publications and book chapters specializing in natural and science-based formulations with enhanced efficacy and consumer desirable performance attributes. Phone: 602-996-9700; E-mail: shyam@biodermresearch.com;
Website: www.biodermresearch.com
References
- Leh et al., Sensors (Basel), 13: 6217–6228 (2013). Published online May 13, 2013. doi: 10.3390/s130506217; Yeo et al., Malaysian Journal of Microbiology, 8:11-20 (2012).
- Kumar et al., Food Chem., 159:451-7 (2014). doi: 10.1016/j.foodchem.2014.03.039. Epub 2014 Mar 20.
- Miao et al., Zhonghua Zheng Xing Wai Ke Za Zhi., 29: 448-52 (2013); www.ncbi.nlm.nih.gov/pubmed/24624885; PLoS One. 2013;8(2):e57226. doi: 10.1371/journal.pone.0057226. Epub 2013 Feb 21; Nigam et al., Chem Biol Interact.,181:77-84 (2009). doi: 10.1016/j.cbi.2009.05.012. Epub 2009 May 27.
- Zin et al., Korean J Physiol Pharmacol.,18:149-53 (2014). doi: 10.4196/kjpp.2014.18.2.149. Epub 2014 Apr 3; Marx et al., BMC Complement Altern Med.,14:134 (2014). doi: 10.1186/1472-6882-14-134.
- Montazeri et al., Iran Red Crescent Med J., 2013 Dec;15(12):e12268. doi: 10.5812/ircmj.12268. Epub 2013 Dec 5; Mandal et al., Pharmacognosy Res., 6:52-7 (2014). doi: 10.4103/0974-8490.122918.
- Mozaffari-Khosravi et al., Complement Ther Med., 22:9-16 (2014). doi: 10.1016/j.ctim.2013.12.017. Epub 2014 Jan 8.
- Chen et al., J Agric Food Chem., 61:1349-56 (2013); doi: 10.1021/jf304340q. Epub 2013 Jan 31; Bhagavathula et al., Wound Repair Regen., 17:360-6 (2009). doi: 10.1111/j.1524-475X.2009.00483.x.
- Sahelian, www.raysahelian.com/kaempferia.html; Park et al., Photodermatol Photoimmunol Photomed. 2013 Dec 9. doi: 10.1111/phpp.12097. [Epub ahead of print].
- Mandawgade et al.,Indian J Pharm Sci., 70: 539-42 (2008). doi: 10.4103/0250-474X.44615.
- Sahu et al., Pak J Biol Sci., 17:146-50 (2014); Mohan et al., Asian Pac J Trop Med., 6:311-4 (2013). doi: 10.1016/S1995-7645(13)60062-0; Sogi et al., Food Chem.,141:2649-55 (2013). doi: 10.1016/j.foodchem.2013.05.053. Epub 2013 May 24; Garcia et al., Plant Foods Hum Nutr., 68:254-8 (2013). doi: 10.1007/s11130-013-0364-y; Ribiero da Silva et al., Food Chem.,143:398-404 (2014). doi: 10.1016/j.foodchem.2013.08.001. Epub 2013 Aug 9.
- Zhao et al., Int J Mol Med., 33:1348-54 (2014). doi: 10.3892/ijmm.2014.1696. Epub 2014 Mar 12; Araujo et al., Molecules,19: 3110-9 (2014). doi: 10.3390/molecules19033110; Ramirez et al., Molecules,19: 438-58 (2013). doi: 10.3390/molecules19010438; Rodeiro et al., Cell Prolif., 47: 48-55 (2014). doi: 10.1111/cpr.12078. Epub 2013 Nov 22; Telang et al., Expert Opin Ther Pat., 23:1561-80 (2013). doi: 10.1517/13543776.2013.836182. Epub 2013 Sep 25; Song et al., Photodermatol Photoimmunol Photomed., 29:84-9 (2013). doi: 10.1111/phpp.12030; Matkowski et al., Mini Rev Med Chem., 3:439-55 (2013).
- Chae et al., Biosci Biotechnol Biochem., 75:2321-5 (2011). Epub 2011 Dec 7.
- Enrique et al., J Environ Manage.,121:202-9 (2013). doi: 10.1016/j.jenvman.2013.02.054. Epub 2013 Mar 29; www.purdue.edu/newsroom/releases/2013/Q4/cellulose-nanocrystals-possible-green-wonder-material.html; http://umaine.edu/pdc/cellulose-nano-crystals/; Marquez et al., Eur J Nutr., 51:729-39 (2012). doi: 10.1007/s00394-011-0252-x. Epub 2011 Oct 7.
- Morffi et al., Plant Foods Hum Nutr., 67:223-8 (2012).
- Ajila et al., Food Chem Toxicol., 48:3406-11 (2010). doi: 10.1016/j.fct.2010.09.012. Epub 2010 Sep 21.
- Lin et al., Eur J Pharmacol. 2014 Apr 18. pii: S0014-2999(14)00290-8. doi: 10.1016/j.ejphar.2014.03.057. [Epub ahead of print]. www.ncbi.nlm.nih.gov/pubmed/24751712 ; Chen et al., Zhongguo Zhong Yao Za Zhi., 39:100-5 (2014). www.ncbi.nlm.nih.gov/pubmed/24754177; Poor et al., J Photochem Photobiol B.,132:1-9 (2014). doi: 10.1016/j.jphotobiol.2014.01.016. Epub 2014 Jan 31; Kim et al., Biol Pharm Bull., 37: 871-876 (2014). Epub 2014 Feb 5; Singh et al., J Food Sci Technol., Jan;51: 67-74 (2014). doi: 10.1007/s13197-011-0479-9. Epub 2011 Aug 17; Hiramitsu et al., Sci Rep., 4:3708 (2014). doi: 10.1038/srep03708; Shakthi Deve et al., J Diabetes Metab Disord.,13:11 (2014). doi: 10.1186/2251-6581-13-11; Kang et al., Biomol Ther (Seoul)., 21:138-45 (2013). doi: 10.4062/biomolther.2013.001. http://www.ncbi.nlm.nih.gov/pubmed/24009872; Jabri Karoui et al., Biomed Res Int., 2013:345415 (2013). doi: 10.1155/2013/345415. Epub 2013 Jun 13. http://www.ncbi.nlm.nih.gov/pubmed/23841062; Barreca et al., Food Chem., 141: 1481-8 (2013). doi: 10.1016/j.foodchem.2013.03.095. Epub 2013 Apr 19.
- Kim et al., Asian Pac J Trop Biomed., 3: 617-22 (2013); discussion 621-2. doi: 10.1016/S2221-1691(13)60125.
- Plaza et al., J Agric Food Chem. 2014 Jan 6. [Epub ahead of print]. www.ncbi.nlm.nih.gov/pubmed/24345041; Panzella et al., Food Chem., 140 :672-9 (2013). doi: 10.1016/j.foodchem.2013.02.121. Epub 2013 Mar 14; Sekhon-Ludoo et al., Food Chem., 140:189-96 (2013). doi: 10.1016/j.foodchem.2013.02.040. Epub 2013 Feb 21; Bai et al., J Sci Food Agric., 93 :2502-6 (2013). doi: 10.1002/jsfa.6066. Epub 2013 Mar 4; Wang et al., J Sep Sci., 36:1652-8 (2013). doi: 10.1002/jssc.201300062. Epub 2013 Apr 23; Thilakarathna et al., Food Chem., 138: 463-70 (2013). doi: 10.1016/j.foodchem.2012.09.121. Epub 2012 Nov 8; Khan et al., Antioxid Redox Signal., 19:151-62 (2013). doi: 10.1089/ars.2012.4901.
- Boyer et al., Nutr J., 3: 5 (2004); Rezk et al., Biochem Biophys Res Commun., 295: 9-13 (2002); van der Sluis et al., J Agric Food Chem., 53:1073-80 (2005); Pearson et al., Life Sci., 64:1913-20 (1999); Chinnici et al., J Agric Food Chem., 52: 4684-9 (2004); Stangl et al., J Nutr., 135:172-8 (2005).
- 20. Manzoor et al., Molecules., 17:390-407 (2012). doi: 10.3390/molecules17010390; Rui et al., Guang Pu Xue Yu Guang Pu Fen Xi., 29:509-10 (2009). www.ncbi.nlm.nih.gov/pubmed/19445239; Cai et al., Arch Gynecol Obstet. 2014 Feb 12. [Epub ahead of print]. www.ncbi.nlm.nih.gov/pubmed/24518938; Mondaini et al., J Sex Med., 6: 2772-7 (2009). doi: 10.1111/j.1743-6109.2009.01393.x. Epub 2009 Jul 21.
- Walia et al., J Sci Food Agric., 94:929-34 (2014). doi: 10.1002/jsfa.6337. Epub 2013 Sep 4.
- Barcia et al., Food Chem., 159:95-105 (2014). doi: 10.1016/j.foodchem.2014.02.163. Epub 2014 Mar 12; Martelo-Vidal et al., Food Chem., 158:28-34 (2014). doi: 10.1016/j.foodchem.2014.02.080. Epub 2014 Feb 28; Papastamoulis et al., J Nat Prod., 77: 213-7 (2014). doi: 10.1021/np4005294. Epub 2014 Feb 12; Fernandez-Marin et al., Food Chem.,135: 1353-9 (2012). doi: 10.1016/j.foodchem.2012.05.086. Epub 2012 May 30; http://en.wikipedia.org/wiki/Phenolic_compounds_in_wine.
- Xuan et al., Acta Pharmacol Sin. 2014 Apr 21. doi: 10.1038/aps.2014.17. [Epub ahead of print]. www.ncbi.nlm.nih.gov/pubmed/24747163.
- Zhang et al., PLoS One., 9: e94302 (2014). doi: 10.1371/journal.pone.0094302. eCollection 2014. www.ncbi.nlm.nih.gov/pubmed/24714160.
- Jin et al., Food Chem Toxicol., 68C:38-43 (2014). doi: 10.1016/j.fct.2014.03.001. [Epub ahead of print].
- Lee et al., Biomol Ther (Seoul)., 22: 35-40 (2014). doi: 10.4062/biomolther.2013.081.