08.27.09
Indication: Cold/Flu
Source: Journal of Applied Research, 2009;9(1&2):30-42.
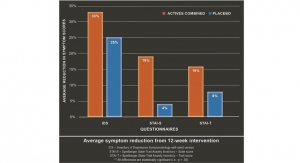
Research: Forty healthy adult subjects (ages 18 to 65) were enrolled in a 12-week randomized, double-blind, placebo-controlled, parallel-group trial conducted during the cold/flu season. The treatment arm compared Wellmune WGP gluco polysaccharide (beta-glucan) (500/mg per day) to a placebo (500 mg rice flour). Cold/flu symptoms were evaluated by medical staff within 24 hours of onset.
Results: There were no significant differences in the incidence of symptomatic respiratory infections (SRIs) among the study groups. However, none of the subjects in the WGP grups missed work or school due to colds, while subjects with colds in the placebo group missed an average of 1.38 days. The WGP group had significantly lower average fever score than the placebo group. No adverse events were detected. According to researchers, this preliminary study suggests 1,3-1,6 beta-glucan from Saccharomyces cerevisiae (Wellmune) may modulate the immune system and reduce some risks associated with upper respiratory influenza infections.