Dr. A. Elizabeth Sloan & Dr. Catherine Adams Hutt, RD03.01.13
While sales of probiotic supplements are projected by Nutrition Business Journal (NBJ) to reach $1 billion in the U.S. by 2013—and Euromonitor estimated global probiotic supplement sales to reach $4 billion by 2016, +48% from 2011; spoonable yogurt to reach $17 billion, +50%; and drinkable yogurt to grow to $21 billion, +50%—it may well be prebiotics, probiotics’ long-overshadowed sidekick, that play the starring role.
Prebiotics are non-digestible food ingredients that beneficially affect the host by selectively stimulating the growth and/or activity of one or a limited number of bacteria in the colon, and thus improve host health. They have a legitimate role in gut health, immunity, bone health and more.
Common prebiotics include inulin, fructo-oligosaccharides (FOS), galacto-oligosaccharides (GOS) and lactulose, which all have a long history of safe use.
NBJ estimated the gastrointestinal (GI) supplement market at $1.5 billion in 2012, +13% vs. 2011. After cholesterol, digestive health was the second most purchased condition-specific food/beverage category in 2011, according to Packaged Facts’ 2012 “Targeted Health & Wellness Foods”; immunity ranked third. Most prebiotics today are carbohydrates and are biochemically active and selectively fermented dietary fibers. “High fiber” was #1 among foods purchased for specific nutritional ingredients in 2011, per Packaged Facts. One in 10 adults (11%) take a fiber supplement, e.g., Metamucil (Mintel, “Attitudes toward Fiber and Digestive Health – US,” March 2012).
Another benefit is reduced adherence of pathogenic bacteria in the gut; and the potential for prebiotics to help prevent or controll infection is an active area of research.
Although most prebiotics today are carbohydrates in nature, they are not required to be, and only need to be able to support the growth of good bacteria and positively affect the health of the host. New and novel prebiotics will likely be the future drivers of the market. Prebiotic’s health benefits include optimal gut health with normal inflammatory response and reduced lower bowel pH, increased mineral absorption (e.g., calcium, magnesium) for optimal bone density and bone health; improved immunity; weight control with greater satiety; and protection from infection.
With awareness of prebiotics at 31% in 2012, according to Multi-Sponsor Surveys’ 2012 Gallup Study of Probiotic & Prebiotic Consumers, it’s time to capitalize not only on the role prebiotics play in supporting “good bacteria,” but also the ability to deliver other high-demand health benefits.
Market Potential
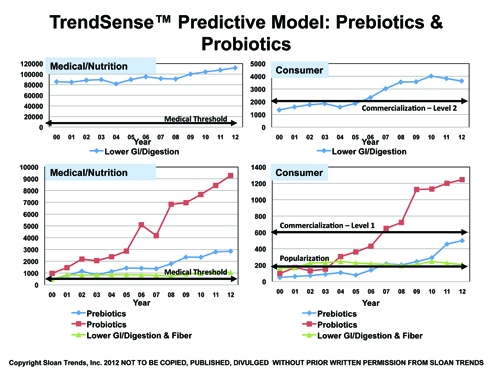
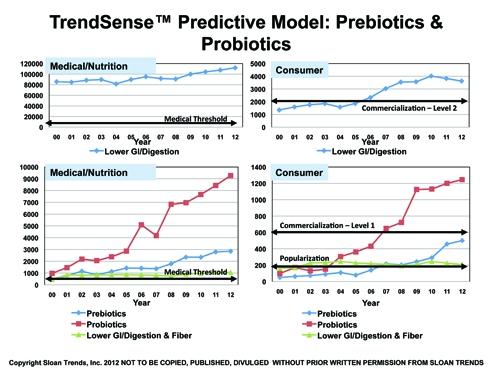
According to Sloan Trends’ TrendSense model, prebiotics are poised to become a mass market opportunity within the next six months. With such fast accelerating Medical Counts, the timing is optimal for launch.
Given the current limited awareness of prebiotic’s specific health benefits, a public relations effort will be essential for success. Prebiotics have been a market opportunity in the health food/specialty channels and among condition-specific/very health conscious consumers since 2007-2008.
In contrast, the marketability of probiotics, which reached mass market status in 2006-2007, continues to grow, although more slowly than earlier in the decade. Supported by a large and growing scientific database, it will remain a solid Level 1 mass market for the foreseeable future.
Lower GI/digestion remains a very strong Level 2 mass market on par with vitamin D and omega 3s, supported by an enormous and fast-accelerating rate of researcher/medical activity.
Fiber/lower GI/digestion is a strong and stable specialty market, but has been lackluster over most of the decade. With dramatic growth over the last five years in Medical Counts, its marketability should have increased. Lack of market emphasis on the lower GI market is the likely culprit.
Growth Opportunities
• The U.S. market for prebiotics is forecast to double in the next five years to more than $220 million (Frost & Sullivan, 2011).
• 9% of consumers are aware of inulin/inulin fiber supplements (Multi-Sponsor 2012).
• 13% are making a strong effort to get more prebiotics in food, beverage or supplement form, 24% some effort; 17% and 34% for probiotics (Multi-Sponsor, 2012).
• 72% of Americans are trying to increase fiber intake; 84% are aware of fiber for weight/satiety, 79% healthy digestion, 43% heart disease, 39% risk of cancer, 36% healthy blood-sugar levels (Packaged Facts’ “Fiber Food Ingredients in the U.S.,” November 2012).
• 76% say it is very important to have a healthy digestive system; 56% of men rate their digestive health as “really good,” compared to 41% of women. (Mintel, March 2012).
• 46% are very/somewhat worried about the long-term effects of GI OTC remedies; 44% becoming too reliant on these products and 40% about their potential negative interactions with medications/vitamins they take (Mintel, “Gastrointestinal Remedies – US,” April 2012).
Regulatory Issues
The regulatory status for carbohydrate prebiotics on the market today is Generally Recognized As Safe (GRAS). It is likely that any novel prebiotic will also be GRAS and exist today in the food supply. Prebiotic compounds introduced as dietary supplements that are not already components of food may require regulatory approval as New Dietary Ingredients (NDI).
The European Union regulates prebiotics as Novel Foods. Prebiotics, including inulin and FOS, were submitted for approval of a health claim but were rejected. Therefore, it is unlikely the European Food Safety Authority (EFSA) will permit any health claim for prebiotics in the near future, just as it has disallowed any health claims for probiotics.
Functional Foods & Beverages
At the present time, interest in prebiotics, as well as successfully launched new products in the U.S., is primarily driven by their ability to help reduce the fat, calorie and sugar content in many foods while maintaining sweetness and adding to the fiber content, rather than an interest in their bifidogenic effects. This could be a missed opportunity. Prebiotics naturally align well with cereals, bars, breads and other higher fiber foods, but are suited for a wide variety of formulations. Euromonitor predicted strong growth for prebiotics in infant formula and milk. Prebiotics can also function as a humectant and mask the bitter notes of bran and high intensity sweeteners. New food/beverages with prebiotics are growing quickly in the global marketplace (291 in 2012 vs. 195 in 2011), holding steady in the U.S. at 61 new products in 2012 (Innova Market Insights, 2013).
Dietary Supplements
While prebiotics appear better suited for foods and beverages due to their functional properties, their health benefits can play a critical role in boosting probiotic and digestive health supplement sales. Prebiotics, alone or combined with probiotics, hold great promise for supplement marketers. Global launches of new prebiotic supplements have fallen dramatically over the past three years, remaining flat in the U.S. with 29 new supplements. GI supplements are among the most trusted categories of dietary supplements, NBJ reported, and marketers should take advantage of the new story of prebiotics.
Dr. A. Elizabeth Sloan and Dr. Catherine Adams Hutt are president and chief scientific and regulatory officer, respectively, of Sloan Trends, Inc., Escondido, CA, a 20-year-old consulting firm that offers trend interpretation/predictions; identifies emerging high potential opportunities; and provides strategic counsel on issues and regulatory claims guidance for food/beverage, supplement and pharmaceutical marketers. For more information: E-mail: lizsloan@sloantrend.com; Website: www.sloantrend.com.
Prebiotics are non-digestible food ingredients that beneficially affect the host by selectively stimulating the growth and/or activity of one or a limited number of bacteria in the colon, and thus improve host health. They have a legitimate role in gut health, immunity, bone health and more.
Common prebiotics include inulin, fructo-oligosaccharides (FOS), galacto-oligosaccharides (GOS) and lactulose, which all have a long history of safe use.
NBJ estimated the gastrointestinal (GI) supplement market at $1.5 billion in 2012, +13% vs. 2011. After cholesterol, digestive health was the second most purchased condition-specific food/beverage category in 2011, according to Packaged Facts’ 2012 “Targeted Health & Wellness Foods”; immunity ranked third. Most prebiotics today are carbohydrates and are biochemically active and selectively fermented dietary fibers. “High fiber” was #1 among foods purchased for specific nutritional ingredients in 2011, per Packaged Facts. One in 10 adults (11%) take a fiber supplement, e.g., Metamucil (Mintel, “Attitudes toward Fiber and Digestive Health – US,” March 2012).
Another benefit is reduced adherence of pathogenic bacteria in the gut; and the potential for prebiotics to help prevent or controll infection is an active area of research.
Although most prebiotics today are carbohydrates in nature, they are not required to be, and only need to be able to support the growth of good bacteria and positively affect the health of the host. New and novel prebiotics will likely be the future drivers of the market. Prebiotic’s health benefits include optimal gut health with normal inflammatory response and reduced lower bowel pH, increased mineral absorption (e.g., calcium, magnesium) for optimal bone density and bone health; improved immunity; weight control with greater satiety; and protection from infection.
With awareness of prebiotics at 31% in 2012, according to Multi-Sponsor Surveys’ 2012 Gallup Study of Probiotic & Prebiotic Consumers, it’s time to capitalize not only on the role prebiotics play in supporting “good bacteria,” but also the ability to deliver other high-demand health benefits.
Market Potential
According to Sloan Trends’ TrendSense model, prebiotics are poised to become a mass market opportunity within the next six months. With such fast accelerating Medical Counts, the timing is optimal for launch.
Given the current limited awareness of prebiotic’s specific health benefits, a public relations effort will be essential for success. Prebiotics have been a market opportunity in the health food/specialty channels and among condition-specific/very health conscious consumers since 2007-2008.
In contrast, the marketability of probiotics, which reached mass market status in 2006-2007, continues to grow, although more slowly than earlier in the decade. Supported by a large and growing scientific database, it will remain a solid Level 1 mass market for the foreseeable future.
Lower GI/digestion remains a very strong Level 2 mass market on par with vitamin D and omega 3s, supported by an enormous and fast-accelerating rate of researcher/medical activity.
Fiber/lower GI/digestion is a strong and stable specialty market, but has been lackluster over most of the decade. With dramatic growth over the last five years in Medical Counts, its marketability should have increased. Lack of market emphasis on the lower GI market is the likely culprit.
Growth Opportunities
• The U.S. market for prebiotics is forecast to double in the next five years to more than $220 million (Frost & Sullivan, 2011).
• 9% of consumers are aware of inulin/inulin fiber supplements (Multi-Sponsor 2012).
• 13% are making a strong effort to get more prebiotics in food, beverage or supplement form, 24% some effort; 17% and 34% for probiotics (Multi-Sponsor, 2012).
• 72% of Americans are trying to increase fiber intake; 84% are aware of fiber for weight/satiety, 79% healthy digestion, 43% heart disease, 39% risk of cancer, 36% healthy blood-sugar levels (Packaged Facts’ “Fiber Food Ingredients in the U.S.,” November 2012).
• 76% say it is very important to have a healthy digestive system; 56% of men rate their digestive health as “really good,” compared to 41% of women. (Mintel, March 2012).
• 46% are very/somewhat worried about the long-term effects of GI OTC remedies; 44% becoming too reliant on these products and 40% about their potential negative interactions with medications/vitamins they take (Mintel, “Gastrointestinal Remedies – US,” April 2012).
Regulatory Issues
The regulatory status for carbohydrate prebiotics on the market today is Generally Recognized As Safe (GRAS). It is likely that any novel prebiotic will also be GRAS and exist today in the food supply. Prebiotic compounds introduced as dietary supplements that are not already components of food may require regulatory approval as New Dietary Ingredients (NDI).
The European Union regulates prebiotics as Novel Foods. Prebiotics, including inulin and FOS, were submitted for approval of a health claim but were rejected. Therefore, it is unlikely the European Food Safety Authority (EFSA) will permit any health claim for prebiotics in the near future, just as it has disallowed any health claims for probiotics.
Functional Foods & Beverages
At the present time, interest in prebiotics, as well as successfully launched new products in the U.S., is primarily driven by their ability to help reduce the fat, calorie and sugar content in many foods while maintaining sweetness and adding to the fiber content, rather than an interest in their bifidogenic effects. This could be a missed opportunity. Prebiotics naturally align well with cereals, bars, breads and other higher fiber foods, but are suited for a wide variety of formulations. Euromonitor predicted strong growth for prebiotics in infant formula and milk. Prebiotics can also function as a humectant and mask the bitter notes of bran and high intensity sweeteners. New food/beverages with prebiotics are growing quickly in the global marketplace (291 in 2012 vs. 195 in 2011), holding steady in the U.S. at 61 new products in 2012 (Innova Market Insights, 2013).
Dietary Supplements
While prebiotics appear better suited for foods and beverages due to their functional properties, their health benefits can play a critical role in boosting probiotic and digestive health supplement sales. Prebiotics, alone or combined with probiotics, hold great promise for supplement marketers. Global launches of new prebiotic supplements have fallen dramatically over the past three years, remaining flat in the U.S. with 29 new supplements. GI supplements are among the most trusted categories of dietary supplements, NBJ reported, and marketers should take advantage of the new story of prebiotics.
Dr. A. Elizabeth Sloan and Dr. Catherine Adams Hutt are president and chief scientific and regulatory officer, respectively, of Sloan Trends, Inc., Escondido, CA, a 20-year-old consulting firm that offers trend interpretation/predictions; identifies emerging high potential opportunities; and provides strategic counsel on issues and regulatory claims guidance for food/beverage, supplement and pharmaceutical marketers. For more information: E-mail: lizsloan@sloantrend.com; Website: www.sloantrend.com.