01.07.20
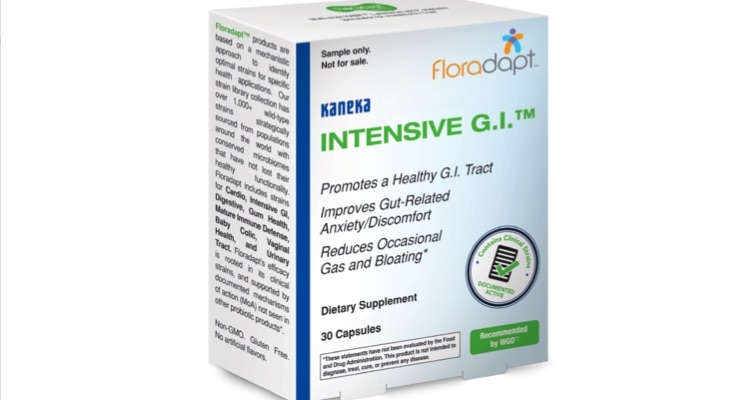
Kaneka Americas Holding, Inc., has achieved self-affirmed Generally Recognized as Safe (GRAS) status in accordance with U.S. FDA safety guidelines for its Intensive G.I. probiotic formulation.
“We are committed to establishing Kaneka’s Floradapt probiotic portfolio as one of the most innovative and efficacious products for a variety of health targets, as supported by scientific evidence,” said Mike Kolifrath, vice president, sales and marketing of the Probiotics Division at Kaneka. “This milestone significantly advances our leadership in the probiotic space and will offer our Intensive G.I. formula access to consumer brands searching for G.I. probiotics with unique mechanisms of action, while removing any doubts on the safety and benefits of the strains.”
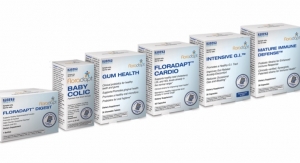
The patented, Non-GMO Floradapt formulations have found success internationally, with distribution in over 60 countries. Intensive G.I. is part of the Floradapt Probiotic Portfolio, which are products based on a mechanistic approach to identify optimal strains for specific health applications. The company’s strain library collection has over 1,000+ strains strategically sourced from populations around the world with conserved microbiomes that have not lost their healthy functionality.
“Safety was demonstrated to the expert panel through a combination of genotypic analysis, phenotypic data, and published clinical trials,” said Fumiki Aoki, business development director, Probiotics Division. “Whole Genome Sequencing was performed to establish the identity of the strains and allow the use of bioinformatics for genomic analysis, which demonstrated the absence of a genetic basis for any safety concerns. Published clinical trials for the strains and species were evaluated for evidence of safety and the absence of adverse effects.”
Floradapt includes multi-strain formulations for Cardio, Digestive, Gum Health, Mature Immune Defense, Baby Colic, Vaginal Health, and Urinary Tract.
“We are committed to establishing Kaneka’s Floradapt probiotic portfolio as one of the most innovative and efficacious products for a variety of health targets, as supported by scientific evidence,” said Mike Kolifrath, vice president, sales and marketing of the Probiotics Division at Kaneka. “This milestone significantly advances our leadership in the probiotic space and will offer our Intensive G.I. formula access to consumer brands searching for G.I. probiotics with unique mechanisms of action, while removing any doubts on the safety and benefits of the strains.”
The patented, Non-GMO Floradapt formulations have found success internationally, with distribution in over 60 countries. Intensive G.I. is part of the Floradapt Probiotic Portfolio, which are products based on a mechanistic approach to identify optimal strains for specific health applications. The company’s strain library collection has over 1,000+ strains strategically sourced from populations around the world with conserved microbiomes that have not lost their healthy functionality.
“Safety was demonstrated to the expert panel through a combination of genotypic analysis, phenotypic data, and published clinical trials,” said Fumiki Aoki, business development director, Probiotics Division. “Whole Genome Sequencing was performed to establish the identity of the strains and allow the use of bioinformatics for genomic analysis, which demonstrated the absence of a genetic basis for any safety concerns. Published clinical trials for the strains and species were evaluated for evidence of safety and the absence of adverse effects.”
Floradapt includes multi-strain formulations for Cardio, Digestive, Gum Health, Mature Immune Defense, Baby Colic, Vaginal Health, and Urinary Tract.