Maged Sharaf, PhD, American Herbal Products Association (AHPA)04.01.16
Like any agricultural commodity, herbs and botanical materials can carry a number of microbes that often originate from the soil and adhere to various parts of the plant. Harvesting, production, manufacturing, transportation and storage practices can also expose herbal ingredients to microbial contaminants. Further, these microorganisms can proliferate depending on a number of factors like moisture and temperature.
Mitigation & Control
The presence of objectionable microorganisms like Escherichia coli, Salmonella spp. and molds can be mitigated by enacting appropriate best practices and controls when producing, harvesting, transporting, storing and manufacturing herbal products. Current good manufacturing practice (cGMP) requirements for dietary supplements establish a comprehensive system of process controls and conditions to protect against potential contamination and proliferation. Documentation requirements at each stage of the manufacturing process also help companies detect problems early, if they arise.
In addition, appropriate testing can help confirm that manufacturing practices are protecting consumers from risks associated with microbial contamination. Food ingredient suppliers, dietary ingredient suppliers and dietary supplement manufacturers ultimately determine what, if any, tests or examinations are appropriate for their ingredients and products.
Microbial tests can be applied to botanical material at various stages of product production and different limits are set according to the intended use of the herbal material. For example, limits can be lower for finished products intended for consumption than for raw plant material intended for further processing, or products intended to be used in boiling water. Limits may also vary for products intended for ingestion versus products intend to be applied topically.
Guidance & Resources
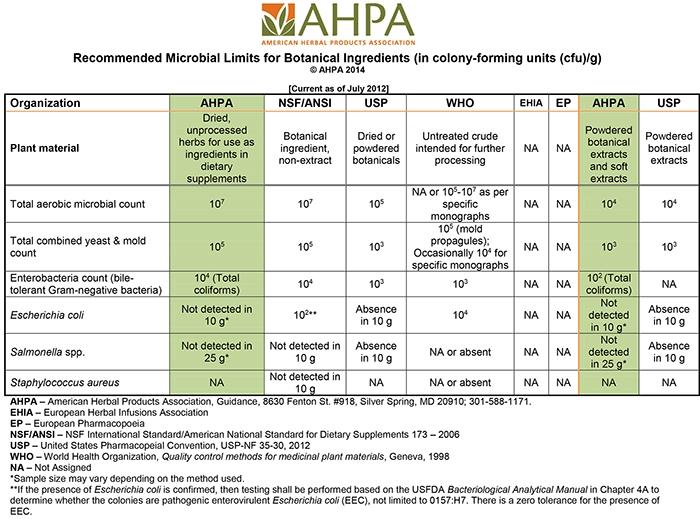
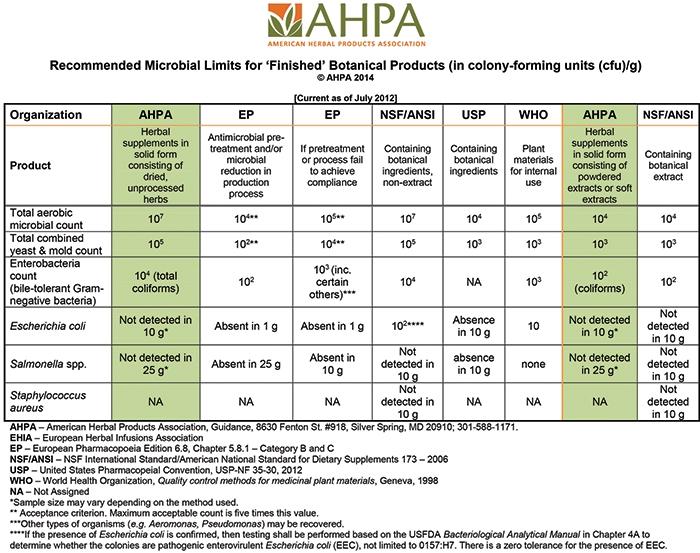
The American Herbal Products Association (AHPA) has developed guidance and resources to help companies understand issues related to microbiology and microbiological characteristics and mycotoxins for which companies may consider testing. AHPA and various regional, national and international standards-setting organizations have set different recommended microbial limits for botanical ingredients and different testing requirements. These can be a useful reference to help companies make the appropriate choice for the selected herbal materials and herbal product. AHPA also provides a table to compare limits set by various organizations.
To reduce the risk of microbial contamination, companies should implement a combination of best practices, controls and documentation. However, there is no one test or best practice that is applicable to every herbal ingredient and supplement. This means companies must weigh numerous factors to develop effective and efficient policies to address the risks associated with microbial contamination.
To help companies understand all the factors and testing that need to be weighed, AHPA recently presented a webinar to help provide the industry with an overview of microbial contamination and to help demystify microbiology testing. The webinar featured industry experts who explained the impact various microorganisms and toxins can have on product integrity, conditions that promote microbial growth and contamination, and strategies to control microorganisms in raw materials and finished products.
The webinar also provided an overview of microbiology testing to prepare attendees for an in-person workshop that will be presented by AHPA and the Joint Institute for Food Safety and Applied Nutrition (JIFSAN) on April 5-7. The workshop will provide hands-on experience with microbiology testing that will help the dietary supplement and herbal product industries ensure product integrity and implement strategies to protect products from microorganisms.
AHPA’s resources provide a good starting point companies can use to help develop policies and procedures or to evaluate their current practices to efficiently reduce the risks associated with microbial contamination.
Maged Sharaf, PhD
American Herbal Products Association (AHPA)
Maged Sharaf, PhD, is the chief science officer of the American Herbal Products Association (AHPA), the national trade association with a focus primarily on herbs and herbal products. AHPA’s membership represents U.S. and international growers, processors, manufacturers, marketers and institutes specializing in botanicals and herbal products. Before AHPA, Dr. Sharaf was the director, Foods, Dietary Supplements and Herbal Medicines at the United States Pharmacopeial Convention (USP). Before USP he was pharmacy associate professor and has preceding experience conducting bioanalytical assay development and validation, and human bioequivalence studies in support of the pharmaceutical industry; and quality control and manufacturing of pharmaceutical dosage forms. He can be reached at 301-588-1171; E-mail: msharaf@ahpa.org; Website: www.ahpa.org.
Mitigation & Control
The presence of objectionable microorganisms like Escherichia coli, Salmonella spp. and molds can be mitigated by enacting appropriate best practices and controls when producing, harvesting, transporting, storing and manufacturing herbal products. Current good manufacturing practice (cGMP) requirements for dietary supplements establish a comprehensive system of process controls and conditions to protect against potential contamination and proliferation. Documentation requirements at each stage of the manufacturing process also help companies detect problems early, if they arise.
In addition, appropriate testing can help confirm that manufacturing practices are protecting consumers from risks associated with microbial contamination. Food ingredient suppliers, dietary ingredient suppliers and dietary supplement manufacturers ultimately determine what, if any, tests or examinations are appropriate for their ingredients and products.
Microbial tests can be applied to botanical material at various stages of product production and different limits are set according to the intended use of the herbal material. For example, limits can be lower for finished products intended for consumption than for raw plant material intended for further processing, or products intended to be used in boiling water. Limits may also vary for products intended for ingestion versus products intend to be applied topically.
Guidance & Resources
The American Herbal Products Association (AHPA) has developed guidance and resources to help companies understand issues related to microbiology and microbiological characteristics and mycotoxins for which companies may consider testing. AHPA and various regional, national and international standards-setting organizations have set different recommended microbial limits for botanical ingredients and different testing requirements. These can be a useful reference to help companies make the appropriate choice for the selected herbal materials and herbal product. AHPA also provides a table to compare limits set by various organizations.
To reduce the risk of microbial contamination, companies should implement a combination of best practices, controls and documentation. However, there is no one test or best practice that is applicable to every herbal ingredient and supplement. This means companies must weigh numerous factors to develop effective and efficient policies to address the risks associated with microbial contamination.
To help companies understand all the factors and testing that need to be weighed, AHPA recently presented a webinar to help provide the industry with an overview of microbial contamination and to help demystify microbiology testing. The webinar featured industry experts who explained the impact various microorganisms and toxins can have on product integrity, conditions that promote microbial growth and contamination, and strategies to control microorganisms in raw materials and finished products.
The webinar also provided an overview of microbiology testing to prepare attendees for an in-person workshop that will be presented by AHPA and the Joint Institute for Food Safety and Applied Nutrition (JIFSAN) on April 5-7. The workshop will provide hands-on experience with microbiology testing that will help the dietary supplement and herbal product industries ensure product integrity and implement strategies to protect products from microorganisms.
AHPA’s resources provide a good starting point companies can use to help develop policies and procedures or to evaluate their current practices to efficiently reduce the risks associated with microbial contamination.
Maged Sharaf, PhD
American Herbal Products Association (AHPA)
Maged Sharaf, PhD, is the chief science officer of the American Herbal Products Association (AHPA), the national trade association with a focus primarily on herbs and herbal products. AHPA’s membership represents U.S. and international growers, processors, manufacturers, marketers and institutes specializing in botanicals and herbal products. Before AHPA, Dr. Sharaf was the director, Foods, Dietary Supplements and Herbal Medicines at the United States Pharmacopeial Convention (USP). Before USP he was pharmacy associate professor and has preceding experience conducting bioanalytical assay development and validation, and human bioequivalence studies in support of the pharmaceutical industry; and quality control and manufacturing of pharmaceutical dosage forms. He can be reached at 301-588-1171; E-mail: msharaf@ahpa.org; Website: www.ahpa.org.