Rebecca Wright02.27.08
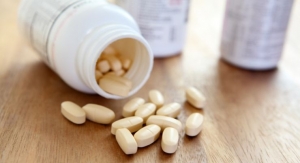
Food supplements should be exempt from the provisions of nutrient profiles because of their small size and dose form, the European Responsible Nutrition Alliance (ERNA) said today.
Food supplements are by definition products presented in dose form, and designed to be taken in measured small quantities to supplement the normal diet. ERNA said that it is essential for manufacturers to be able to communicate on their intended use and beneficial properties to the consumer – communications that are considered health claims.
“Typically, food supplements are marketed in the form of capsules, tablets, pills, and represent a maximum weight of two grams,” said Gert Krabichler, chair of ERNA. “Such forms do not contain significant quantities of energy, fat, sugar, and salt, and therefore do not add to the daily energy-intake of the consumer. The reasons for imposing nutrient profiles therefore are irrelevant for food supplements.”
The opinion of EFSA which was released this week, although it mentions the possibility of exemptions, does not cover such exemption explicitly for food supplements, which is surprising as this was discussed extensively at the EFSA colloquium on Nutrient profiles last November in Parma and there seemed to be general agreement.
ERNA has appealed to the European Commission to consider such exemption, saying that food supplements generally contribute less than 25kcal per day, per daily recommended amount, which is insignificant and should not lead to restrictions for making claims, since nutrient profiles are intended to restrict the possibility of health claims on foods that contribute substantially to the intake of fat, sugar, salt.
Mr Krabichler said: “If no account is taken of the specificities of food supplements when establishing nutrient profiles, the result could be that certain food supplements would no longer be allowed to make health claims, while regular foods that contribute substantially more of these nutrients in terms of daily intake would still be permitted to make claims.”
Food supplements are by definition products presented in dose form, and designed to be taken in measured small quantities to supplement the normal diet. ERNA said that it is essential for manufacturers to be able to communicate on their intended use and beneficial properties to the consumer – communications that are considered health claims.
“Typically, food supplements are marketed in the form of capsules, tablets, pills, and represent a maximum weight of two grams,” said Gert Krabichler, chair of ERNA. “Such forms do not contain significant quantities of energy, fat, sugar, and salt, and therefore do not add to the daily energy-intake of the consumer. The reasons for imposing nutrient profiles therefore are irrelevant for food supplements.”
The opinion of EFSA which was released this week, although it mentions the possibility of exemptions, does not cover such exemption explicitly for food supplements, which is surprising as this was discussed extensively at the EFSA colloquium on Nutrient profiles last November in Parma and there seemed to be general agreement.
ERNA has appealed to the European Commission to consider such exemption, saying that food supplements generally contribute less than 25kcal per day, per daily recommended amount, which is insignificant and should not lead to restrictions for making claims, since nutrient profiles are intended to restrict the possibility of health claims on foods that contribute substantially to the intake of fat, sugar, salt.
Mr Krabichler said: “If no account is taken of the specificities of food supplements when establishing nutrient profiles, the result could be that certain food supplements would no longer be allowed to make health claims, while regular foods that contribute substantially more of these nutrients in terms of daily intake would still be permitted to make claims.”