A. Elizabeth Sloan and Catherine Adams Hutt07.01.12
There’s no doubt consumers are looking for the fastest acting, most effective and most cost-effective supplement possible. Choosing the right form can help to convey these messages and serve as a major product differentiator.
Effectiveness & Performance
“Expanded/improved effectiveness” is among the top claims that appeared on the best-selling new healthcare products—including supplements, over-the-counter (OTC) drugs, beauty and personal care products—in 2011 (see Figure 1)1.
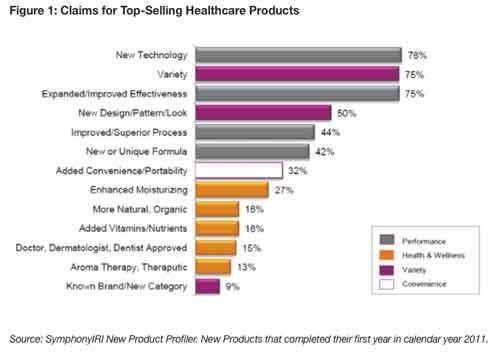
While the horsepower race for better performing products is not new, what is new is that consumers are embracing technology to reach this goal. Three-quarters (76%) of last year’s top-selling new healthcare products carried a new technology claim; 44% “improved/superior process” and 42% “a new or unique formula”1.
One-third (32%) of these products touted “added convenience/portability”; 16% claimed more “natural or organic”; and 15% “doctor, dermatologist or dentist approved”1.
Form is virtually an untapped tool when it comes to attracting the 74% of consumers who are looking for multi-functional supplements/OTCs that address more than one need or symptom2.
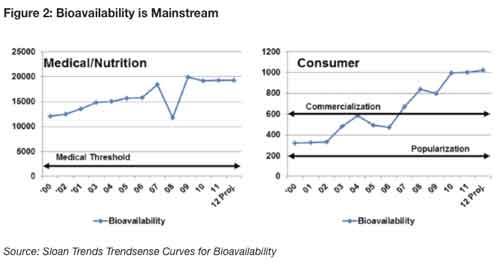
Form can also play a key role in enhancing bioavailability, which became a mass-market opportunity in 2007 and has escalated ever since3. The Hartman Group, Bellevue, WA, reports that core supplement users are limiting the number of supplements they take daily due to pervasive doubts about bioavailability4. One-third (34%) of supplement marketers are actively working to improve bioavailability(see Figure 2)5.
Clean labels and purity remain top selection factors for 42% of supplement/OTC users; lack of chemicals/irritants for 31%. Form selection must take these consumer desires into account6. Natural, gluten-free, vegetarian, no preservatives and no artificial colors/flavors top the list of claims on vitamin/mineral supplements7. Half (52%) of consumers avoid artificial ingredients, 47% artificial flavors and 42% artificial colors8. Nearly 10% of new global gummy launches in 2011 were gelatin-free9.
Preferred Pill Forms
Despite the current hype that supplement users are frantically looking for new delivery systems, Hartman reports that users overwhelmingly prefer supplements in “pill” form8.
While tablets remain the most used pill supplement form by 60% of users, consumer preferences have changed dramatically since 2002. Liquid-filled/softgels are preferred by 65% of consumers, gelcoat/softlets 60%, capsules 57% and caplets 47%; preference for tablets fell from 57% in 2002 to 43% in 2009, and from 55% to 42% for chewable tablets10.
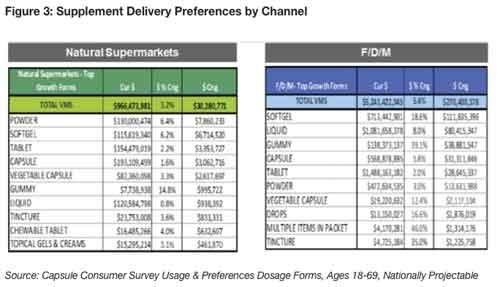
According to SPINS, sales of softgels in mass channels grew more than 18% and 6% in natural supermarkets; while capsules were up nearly 6% and up 2%, respectively11. Tablets remain the largest sector in mass channels, while capsules continue to rule the natural channel (see Figure 3)10.
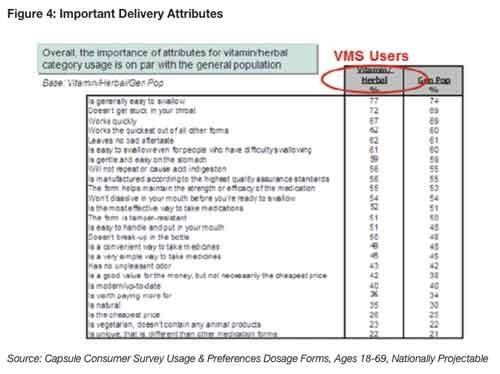
“Easy-to-swallow” and “doesn’t get stuck in the throat” are the most important dosage/form criteria for 77% and 72% of supplement users, respectively, according to a Capsugel consumer survey10. Forty-four percent of adults have experienced difficulty at one time or another trying to swallow pills; 14% alerted a doctor to their difficulty12.
“Works quickly,” “works the quickest of all forms,” “no bad aftertaste” and “gentle on the stomach” were other criteria ranked among the top two choices by 60% or more of users (see Figure 4)10.
“Very important”/”important” form attributes include the following for more than half of supplement users10:
• Will not repeat or cause acid indigestion
• Manufactured by the highest quality assurance standards
• The form helps maintain the strength or efficacy of the medication
• Won’t dissolve in your mouth before you’re ready to swallow it
• Is the most effective way to take medications
• Tamper resistant
• Is easy to handle and put in your mouth
• Doesn’t break up in the bottle
Price was one of the lowest ranking supplement dose/form attributes, with only 26% of supplement users listing it among their top two “very important”/”important” attributes. “A good value for the money, but not necessarily the cheapest price” was cited by 42% of users as “very”/”somewhat important”10.
Supplement users closely associate capsules with “easy to swallow”; gelcoats are associated with both “easy to swallow” and “no bad aftertaste”; liquid-filled are associated with being “quickest”; and tablets are most associated with the “cheapest price”11.
Mini’s, petites, pearls and quick-release tablets are high potential forms that are well positioned in the quick-release, easy to swallow and fast-acting segment.
Perhaps most exciting, however, are the new colorful, multi-component capsules, e.g., Capsugel's beads and liquid—or capsules within a capsule—delivery systems, which provide supplement users with a unique visual presentation for multi-release, quick releases and targeted release concepts.
Likewise, bi- and tri-layered colored tablets, enteric coatings, acid resistant coatings and microencapsulated nutrients can all deliver unique functionalities for more than one ingredient, while supporting bioavailability. Some technologies can ensure survival of the active substances through the high acid environment of the stomach with targeted delivery to the intestine for optimal absorption.
Biodegradable polymers, star polymers, dendrimers and Buckyballs/C-60 Fullerene are among the next generation of delivery systems for supplements, crossing over from the pharmaceutical industry.
The future of the pill form of supplement is strong with older Americans. One-third of all Americans are now age 55 or older and they are the heaviest supplement users. They equate dietary supplement pills with “medical effectiveness” and are the least likely to use functional and fortified foods/beverages7.
Liquidfication
Liquid forms represent one of the fastest growing supplement sectors. SPINS reports that liquid forms are among the largest supplement sections in both natural and mass channels—up 8% in mass channels, but exhibiting somewhat lackluster growth in the natural channel. On the other hand, powders posted respectable growth in the natural channel, with gains more tempered in the mass market (see Figure 3)11.
Weight control/nutrition liquids/powders and energy drinks were both up 18% in unit sales in 2011; followed by sports drinks, up 11%. These categories were the fastest growing consumer product goods in 2011 for the food, drug, mass, and convenience stores (excluding Walmart)13.
In May 2011, U.S. energy shot sales crossed the $1 billion mark in food, drug, mass merchandiser and convenience stores, up nearly 34% over the previous year14. One-third (31%) of consumers aged 18-24 years old drank an energy shot at least once a month, 26% aged 25-35, 19% aged 35-44 and 11% aged 45-5415.
Energy shots were the fastest growing category in convenience stores in 2011 with sales up 36%; supplement sales reached almost $2 billion in C-stores16. Energy was among the top reason for use of shots—there was no mention of convenience or added potency, suggesting that the end benefit, not the form, is responsible for their high appeal17.
Shots now deliver a wide variety of end benefits, including energy, inner beauty, hangover relief and enhanced sexual performance. They are also being marketed by mainstream manufacturers and boutique marketers—note the launch of Campbell’s V8 V-Fusion+Energy shots.
Shots and liquids allow the supplement market to take advantage of flavor preferences. Tum’s antacids have enjoyed great success by flavoring its chewable tablets; sales of Tum’s in mass channels rose 26% in 201118.
SymphonyIRI data for the year ending August 15, 2011 shows a slightly less robust picture for powdered drinks in mass channels. Crystal Light on the Go Pure Fitness enjoyed strong growth, albeit from a very small base. Alacer Emergen-C ranked 25th among the top “niche brands” in drug stores with sales of $67 million; Airborne 28th with sales of $64 million in drug stores19.
GoodBelly ToGo Probiotic Sticks and SlimStyles PgX SlimStyx are two popular powdered supplements. With sales of bottled water up 4% for the first time in several years, effervescent options and powders may find a more welcome market13.
Sports nutrition powders are by far the major driver in the sports supplement category, with sales projected to grow from $2.8 billion in 2010 to $4.2 billion by 201720.
Highly potent pure liquid drops are considered good delivery systems for vitamins and minerals, as are effervescents. Tinctures grew 35% in mass-market supplement sales, albeit from a very small base.
Vials represent another hot trend emanating from Europe. U.S. sales of nutrition bars, gels, chews and other innovative delivery forms were up 10% in 2010 to $2.5 billion21.
Liquids can deliver a positive story for enhanced bioavailability, since absorption can begin immediately in the mucosal lining of the mouth. However, like other supplements, nutrients must be protected so that they may pass through the acid environment of the stomach and be delivered to their target for primary absorption in the small intestine.
Of particular note, this segment will be carefully watched by FDA and FTC. It is a fertile area for abuse in combination with alcohol and is targeted to the youth market. Avoiding these regulatory pitfalls will be important to the viability of the liquid shot market for the future.
Candy Forms are Sweet!
Gummies are among the fastest growing supplement forms, according to SPINS, with sales up 38% in mass channels to $138 million, and nearly 15% in the natural channel11. Globally, gummy candy is the third most active new product candy category, following chocolate and hard candy9. “Better-for-you” is the leading gummy candy segment in the U.S. The overall gummy candy segment was estimated at $900 million in 20119,22.
For the year ending March 18, 2012, Flintstone’s ranked 5th in the multivitamin category in drug stores, with unit sales up 4.6%; One-A-Day Vita Craves 10th, up 2%23.
Claims including a “full serving of fruit/vegetables,” “high potency” and “natural functionality” (e.g., naturally high in antioxidants from juice) are among the new gummy positionings. Forty-five percent of consumers prefer to see “made with real fruit” on candies9. Other new consumer-preferred identities include “made with real fruit pulp,” “yogurt gummies,” “liquid-filled gummy bursts,” “jelly beans” and “green tea added.” Mars/Wrigley is leading in the liquid-filled gummy candy segment with sales of $225 million22. Chews are also increasing in popularity as a supplement form.
There are regulatory constraints for gummy type supplements, because they may so closely resemble candy. FDA has a policy against fortifying “snack” foods and “candy.” In fact, it sent a warning letter to Coke when Diet Coke Plus with B vitamins was launched.
Although still only a small segment of the U.S. $3.7 billion gum market, functional gums hold high potential for dietary supplements, as 58% of American consumers chew gum24.
Kraft Foods Inc.'s Trident Vitality line offers Vigorate, Rejuve and Awaken, with vitamin C, ginseng or white tea infused into each gum pellet. Wrigley’s 5, by Wm. Wrigley Jr. Co, owned by Mars Inc., markets React, Zing and Flare.
Science ties gum as a form to four major health categories: improved oral health, cravings control/weight, mental focus/alertness and stress relief. Moreover, gum is an excellent nutrient delivery method for nutrient absorption in the mouth, similar to sublinguals in principle25.
Four in 10 (44%) consumers have not tried gum for weight control, but would like to do so; 48% of those 45-54 and 44% aged 55-6424. More than half (54%) of consumers would like to try gum with antioxidants. Lotte in Japan sells “stress gums” and “gums with collagen” for beauty9.
Energizing gums are of interest to one-third of consumers, skewing higher to those aged 18-3724. Since World War I, the U.S. Armed Forces has supplied chewing gum to the field and in combat rations.
Gum also plays an important role for athletes—59% of everyday athletes chew gum to help focus on their sporting event; 58% of college athletes25.
Be Careful of Some Emerging Forms
There are several supplement forms that may be on the market, but should be avoided due to regulatory constraints and the potential for regulatory action. Sublinguals, dissolvable strips, nasal sprays and inhalers are being used as forms for dietary supplements. Products intended to be administered sublingually or through inhalation are not considered to be dietary supplements by FDA.
A dietary supplement is a product intended to supplement the diet. Dietary supplements must be ingested into the stomach and gastrointestinal tract by means of enteral administration. Only products that are intended for ingestion may be lawfully marketed as dietary supplements. Products intended to enter the body directly through the skin or mucosal tissues are not dietary supplements. A sublingual product or inhaled supplement will be considered a drug by FDA if it is designed to deliver a substance that influences the structure or function of the body.
In Perfect Form for the Future
Form is an integral part of delivering effective end benefits for dietary supplements. While timing and regulatory challenges may be less than optimal for some of the more novel and drug-like delivery systems, there are a plethora of new concepts, from pills to great tasting playful forms and formulations such as effervescents and gummies.
Moreover, many of today’s consumers have deeply ingrained ideas about how certain micronutrients should be packaged that may be difficult to reshape, e.g., omega 3 supplements should come in gelcaps, high potency vitamins in large tablets. That said, consumers view multivitamins lightly and will likely be receptive to new forms. This is an excellent place to start.
About the author: Dr. A. Elizabeth Sloan is president of Sloan Trends, Inc., Escondido, CA, which is a consulting firm that offers trend-tracking and predictions, strategic counsel and business-building ideas for food, supplement and pharmaceutical marketers. She can be reached at lizsloan@sloantrend.com
1. SymphonyIRI, 2012. 2011 “New Product Pacesetters.” Times & Trends, April. SymphonyIRI Group, Chicago, IL. www.infores.com..
2. SymphonyIRI, 2011. 2010 “New Product Pacesetters.” Times & Trends, March.
3. Sloan Trends, 2012. TrendSense Report on Nutrient Bioavailability. Sloan Trends, Inc. Escondido, CA. www.sloantrend.com.
4. Hartman, 2010. “Reimagining Health & Nutrition.” The Hartman Group, Bellevue, WA.www.hartman-group.com.
5. NBJ. 2011. XVI(9).
6. Anon. 2010. Chain Drug Review, 3/1/10; p. 36.
7. Packaged Facts, “Nutritional Supplements in the U.S., 2010”
8. Hartman Deep Dive
9. Innova Market Insights, 2012.
10. “Capsugel Consumer Survey Usage & Preferences Dosage Forms,” 2009.
11. SPINS, 2011. SPINSscan Natural Excl. Whole Foods and SPINSscan FDM Conventional Powered By Nielsen Scantrack-52 Weeks Ending 12-25-10
12. Louis Harris, 2009. “2009 Healthcare Survey.”
13. IRI. 2012. CPC “2011year in review.” Times & Trends, Feb.
14. IRI. 2011c. Press release, May 15.
15. Sprinkle, D. 2012. “Innovation Consumers Want.” Presented at Nutracon, Anaheim, Calif., March 7-8.
16. CSNews. 2012. Convenience operator’s survey. Convenience Store News. www.csnews.com.
17. Mintel. 2011. “Energy Drinks & Energy Shots-US.” Mintel International. Chicago, IL.
18. SymphonyIRI, Year ended 8/2011.
19. NBJ. 2011a. Sales data sheets 2011. Nutr. Bus. J., Boulder, Colo. www.nutritionbusiness.com.
20. Alexander, A. 2012. Niche Brands. Drug Store News 34(7): 24-36.
21. NBJ, 2012. Top U.S. Nutrition Bars, Energy Gels and Chews. www.Nutritionbusiness.com.
22. Candy Manufacturers of America. Sales data for 2011, SymphonyIRI.
23. Anon., 2012. “OTC Sales in Drug Stores by Category.” Chain Drug Review. 34(9): 25-45.
24. Mintel, 2009. “Candy, Gum and Mints – U.S.” Mintel International. Chicago, IL. www.mintel.com.
25. Wrigley Institute (link found at www.wrigleys.com).
Effectiveness & Performance
“Expanded/improved effectiveness” is among the top claims that appeared on the best-selling new healthcare products—including supplements, over-the-counter (OTC) drugs, beauty and personal care products—in 2011 (see Figure 1)1.
While the horsepower race for better performing products is not new, what is new is that consumers are embracing technology to reach this goal. Three-quarters (76%) of last year’s top-selling new healthcare products carried a new technology claim; 44% “improved/superior process” and 42% “a new or unique formula”1.
One-third (32%) of these products touted “added convenience/portability”; 16% claimed more “natural or organic”; and 15% “doctor, dermatologist or dentist approved”1.
Form is virtually an untapped tool when it comes to attracting the 74% of consumers who are looking for multi-functional supplements/OTCs that address more than one need or symptom2.
Form can also play a key role in enhancing bioavailability, which became a mass-market opportunity in 2007 and has escalated ever since3. The Hartman Group, Bellevue, WA, reports that core supplement users are limiting the number of supplements they take daily due to pervasive doubts about bioavailability4. One-third (34%) of supplement marketers are actively working to improve bioavailability(see Figure 2)5.
Clean labels and purity remain top selection factors for 42% of supplement/OTC users; lack of chemicals/irritants for 31%. Form selection must take these consumer desires into account6. Natural, gluten-free, vegetarian, no preservatives and no artificial colors/flavors top the list of claims on vitamin/mineral supplements7. Half (52%) of consumers avoid artificial ingredients, 47% artificial flavors and 42% artificial colors8. Nearly 10% of new global gummy launches in 2011 were gelatin-free9.
Preferred Pill Forms
Despite the current hype that supplement users are frantically looking for new delivery systems, Hartman reports that users overwhelmingly prefer supplements in “pill” form8.
While tablets remain the most used pill supplement form by 60% of users, consumer preferences have changed dramatically since 2002. Liquid-filled/softgels are preferred by 65% of consumers, gelcoat/softlets 60%, capsules 57% and caplets 47%; preference for tablets fell from 57% in 2002 to 43% in 2009, and from 55% to 42% for chewable tablets10.
According to SPINS, sales of softgels in mass channels grew more than 18% and 6% in natural supermarkets; while capsules were up nearly 6% and up 2%, respectively11. Tablets remain the largest sector in mass channels, while capsules continue to rule the natural channel (see Figure 3)10.
“Easy-to-swallow” and “doesn’t get stuck in the throat” are the most important dosage/form criteria for 77% and 72% of supplement users, respectively, according to a Capsugel consumer survey10. Forty-four percent of adults have experienced difficulty at one time or another trying to swallow pills; 14% alerted a doctor to their difficulty12.
“Works quickly,” “works the quickest of all forms,” “no bad aftertaste” and “gentle on the stomach” were other criteria ranked among the top two choices by 60% or more of users (see Figure 4)10.
“Very important”/”important” form attributes include the following for more than half of supplement users10:
• Will not repeat or cause acid indigestion
• Manufactured by the highest quality assurance standards
• The form helps maintain the strength or efficacy of the medication
• Won’t dissolve in your mouth before you’re ready to swallow it
• Is the most effective way to take medications
• Tamper resistant
• Is easy to handle and put in your mouth
• Doesn’t break up in the bottle
Price was one of the lowest ranking supplement dose/form attributes, with only 26% of supplement users listing it among their top two “very important”/”important” attributes. “A good value for the money, but not necessarily the cheapest price” was cited by 42% of users as “very”/”somewhat important”10.
Supplement users closely associate capsules with “easy to swallow”; gelcoats are associated with both “easy to swallow” and “no bad aftertaste”; liquid-filled are associated with being “quickest”; and tablets are most associated with the “cheapest price”11.
Mini’s, petites, pearls and quick-release tablets are high potential forms that are well positioned in the quick-release, easy to swallow and fast-acting segment.
Perhaps most exciting, however, are the new colorful, multi-component capsules, e.g., Capsugel's beads and liquid—or capsules within a capsule—delivery systems, which provide supplement users with a unique visual presentation for multi-release, quick releases and targeted release concepts.
Likewise, bi- and tri-layered colored tablets, enteric coatings, acid resistant coatings and microencapsulated nutrients can all deliver unique functionalities for more than one ingredient, while supporting bioavailability. Some technologies can ensure survival of the active substances through the high acid environment of the stomach with targeted delivery to the intestine for optimal absorption.
Biodegradable polymers, star polymers, dendrimers and Buckyballs/C-60 Fullerene are among the next generation of delivery systems for supplements, crossing over from the pharmaceutical industry.
The future of the pill form of supplement is strong with older Americans. One-third of all Americans are now age 55 or older and they are the heaviest supplement users. They equate dietary supplement pills with “medical effectiveness” and are the least likely to use functional and fortified foods/beverages7.
Liquidfication
Liquid forms represent one of the fastest growing supplement sectors. SPINS reports that liquid forms are among the largest supplement sections in both natural and mass channels—up 8% in mass channels, but exhibiting somewhat lackluster growth in the natural channel. On the other hand, powders posted respectable growth in the natural channel, with gains more tempered in the mass market (see Figure 3)11.
Weight control/nutrition liquids/powders and energy drinks were both up 18% in unit sales in 2011; followed by sports drinks, up 11%. These categories were the fastest growing consumer product goods in 2011 for the food, drug, mass, and convenience stores (excluding Walmart)13.
In May 2011, U.S. energy shot sales crossed the $1 billion mark in food, drug, mass merchandiser and convenience stores, up nearly 34% over the previous year14. One-third (31%) of consumers aged 18-24 years old drank an energy shot at least once a month, 26% aged 25-35, 19% aged 35-44 and 11% aged 45-5415.
Energy shots were the fastest growing category in convenience stores in 2011 with sales up 36%; supplement sales reached almost $2 billion in C-stores16. Energy was among the top reason for use of shots—there was no mention of convenience or added potency, suggesting that the end benefit, not the form, is responsible for their high appeal17.
Shots now deliver a wide variety of end benefits, including energy, inner beauty, hangover relief and enhanced sexual performance. They are also being marketed by mainstream manufacturers and boutique marketers—note the launch of Campbell’s V8 V-Fusion+Energy shots.
Shots and liquids allow the supplement market to take advantage of flavor preferences. Tum’s antacids have enjoyed great success by flavoring its chewable tablets; sales of Tum’s in mass channels rose 26% in 201118.
SymphonyIRI data for the year ending August 15, 2011 shows a slightly less robust picture for powdered drinks in mass channels. Crystal Light on the Go Pure Fitness enjoyed strong growth, albeit from a very small base. Alacer Emergen-C ranked 25th among the top “niche brands” in drug stores with sales of $67 million; Airborne 28th with sales of $64 million in drug stores19.
GoodBelly ToGo Probiotic Sticks and SlimStyles PgX SlimStyx are two popular powdered supplements. With sales of bottled water up 4% for the first time in several years, effervescent options and powders may find a more welcome market13.
Sports nutrition powders are by far the major driver in the sports supplement category, with sales projected to grow from $2.8 billion in 2010 to $4.2 billion by 201720.
Highly potent pure liquid drops are considered good delivery systems for vitamins and minerals, as are effervescents. Tinctures grew 35% in mass-market supplement sales, albeit from a very small base.
Vials represent another hot trend emanating from Europe. U.S. sales of nutrition bars, gels, chews and other innovative delivery forms were up 10% in 2010 to $2.5 billion21.
Liquids can deliver a positive story for enhanced bioavailability, since absorption can begin immediately in the mucosal lining of the mouth. However, like other supplements, nutrients must be protected so that they may pass through the acid environment of the stomach and be delivered to their target for primary absorption in the small intestine.
Of particular note, this segment will be carefully watched by FDA and FTC. It is a fertile area for abuse in combination with alcohol and is targeted to the youth market. Avoiding these regulatory pitfalls will be important to the viability of the liquid shot market for the future.
Candy Forms are Sweet!
Gummies are among the fastest growing supplement forms, according to SPINS, with sales up 38% in mass channels to $138 million, and nearly 15% in the natural channel11. Globally, gummy candy is the third most active new product candy category, following chocolate and hard candy9. “Better-for-you” is the leading gummy candy segment in the U.S. The overall gummy candy segment was estimated at $900 million in 20119,22.
For the year ending March 18, 2012, Flintstone’s ranked 5th in the multivitamin category in drug stores, with unit sales up 4.6%; One-A-Day Vita Craves 10th, up 2%23.
Claims including a “full serving of fruit/vegetables,” “high potency” and “natural functionality” (e.g., naturally high in antioxidants from juice) are among the new gummy positionings. Forty-five percent of consumers prefer to see “made with real fruit” on candies9. Other new consumer-preferred identities include “made with real fruit pulp,” “yogurt gummies,” “liquid-filled gummy bursts,” “jelly beans” and “green tea added.” Mars/Wrigley is leading in the liquid-filled gummy candy segment with sales of $225 million22. Chews are also increasing in popularity as a supplement form.
There are regulatory constraints for gummy type supplements, because they may so closely resemble candy. FDA has a policy against fortifying “snack” foods and “candy.” In fact, it sent a warning letter to Coke when Diet Coke Plus with B vitamins was launched.
Although still only a small segment of the U.S. $3.7 billion gum market, functional gums hold high potential for dietary supplements, as 58% of American consumers chew gum24.
Kraft Foods Inc.'s Trident Vitality line offers Vigorate, Rejuve and Awaken, with vitamin C, ginseng or white tea infused into each gum pellet. Wrigley’s 5, by Wm. Wrigley Jr. Co, owned by Mars Inc., markets React, Zing and Flare.
Science ties gum as a form to four major health categories: improved oral health, cravings control/weight, mental focus/alertness and stress relief. Moreover, gum is an excellent nutrient delivery method for nutrient absorption in the mouth, similar to sublinguals in principle25.
Four in 10 (44%) consumers have not tried gum for weight control, but would like to do so; 48% of those 45-54 and 44% aged 55-6424. More than half (54%) of consumers would like to try gum with antioxidants. Lotte in Japan sells “stress gums” and “gums with collagen” for beauty9.
Energizing gums are of interest to one-third of consumers, skewing higher to those aged 18-3724. Since World War I, the U.S. Armed Forces has supplied chewing gum to the field and in combat rations.
Gum also plays an important role for athletes—59% of everyday athletes chew gum to help focus on their sporting event; 58% of college athletes25.
Be Careful of Some Emerging Forms
There are several supplement forms that may be on the market, but should be avoided due to regulatory constraints and the potential for regulatory action. Sublinguals, dissolvable strips, nasal sprays and inhalers are being used as forms for dietary supplements. Products intended to be administered sublingually or through inhalation are not considered to be dietary supplements by FDA.
A dietary supplement is a product intended to supplement the diet. Dietary supplements must be ingested into the stomach and gastrointestinal tract by means of enteral administration. Only products that are intended for ingestion may be lawfully marketed as dietary supplements. Products intended to enter the body directly through the skin or mucosal tissues are not dietary supplements. A sublingual product or inhaled supplement will be considered a drug by FDA if it is designed to deliver a substance that influences the structure or function of the body.
In Perfect Form for the Future
Form is an integral part of delivering effective end benefits for dietary supplements. While timing and regulatory challenges may be less than optimal for some of the more novel and drug-like delivery systems, there are a plethora of new concepts, from pills to great tasting playful forms and formulations such as effervescents and gummies.
Moreover, many of today’s consumers have deeply ingrained ideas about how certain micronutrients should be packaged that may be difficult to reshape, e.g., omega 3 supplements should come in gelcaps, high potency vitamins in large tablets. That said, consumers view multivitamins lightly and will likely be receptive to new forms. This is an excellent place to start.
About the author: Dr. A. Elizabeth Sloan is president of Sloan Trends, Inc., Escondido, CA, which is a consulting firm that offers trend-tracking and predictions, strategic counsel and business-building ideas for food, supplement and pharmaceutical marketers. She can be reached at lizsloan@sloantrend.com
References
1. SymphonyIRI, 2012. 2011 “New Product Pacesetters.” Times & Trends, April. SymphonyIRI Group, Chicago, IL. www.infores.com..
2. SymphonyIRI, 2011. 2010 “New Product Pacesetters.” Times & Trends, March.
3. Sloan Trends, 2012. TrendSense Report on Nutrient Bioavailability. Sloan Trends, Inc. Escondido, CA. www.sloantrend.com.
4. Hartman, 2010. “Reimagining Health & Nutrition.” The Hartman Group, Bellevue, WA.www.hartman-group.com.
5. NBJ. 2011. XVI(9).
6. Anon. 2010. Chain Drug Review, 3/1/10; p. 36.
7. Packaged Facts, “Nutritional Supplements in the U.S., 2010”
8. Hartman Deep Dive
9. Innova Market Insights, 2012.
10. “Capsugel Consumer Survey Usage & Preferences Dosage Forms,” 2009.
11. SPINS, 2011. SPINSscan Natural Excl. Whole Foods and SPINSscan FDM Conventional Powered By Nielsen Scantrack-52 Weeks Ending 12-25-10
12. Louis Harris, 2009. “2009 Healthcare Survey.”
13. IRI. 2012. CPC “2011year in review.” Times & Trends, Feb.
14. IRI. 2011c. Press release, May 15.
15. Sprinkle, D. 2012. “Innovation Consumers Want.” Presented at Nutracon, Anaheim, Calif., March 7-8.
16. CSNews. 2012. Convenience operator’s survey. Convenience Store News. www.csnews.com.
17. Mintel. 2011. “Energy Drinks & Energy Shots-US.” Mintel International. Chicago, IL.
18. SymphonyIRI, Year ended 8/2011.
19. NBJ. 2011a. Sales data sheets 2011. Nutr. Bus. J., Boulder, Colo. www.nutritionbusiness.com.
20. Alexander, A. 2012. Niche Brands. Drug Store News 34(7): 24-36.
21. NBJ, 2012. Top U.S. Nutrition Bars, Energy Gels and Chews. www.Nutritionbusiness.com.
22. Candy Manufacturers of America. Sales data for 2011, SymphonyIRI.
23. Anon., 2012. “OTC Sales in Drug Stores by Category.” Chain Drug Review. 34(9): 25-45.
24. Mintel, 2009. “Candy, Gum and Mints – U.S.” Mintel International. Chicago, IL. www.mintel.com.
25. Wrigley Institute (link found at www.wrigleys.com).