05.15.12
BCM-95, a patented, high bioavailability curcumin, was found to be equal to the prescription rheumatoid arthritis drug, diclofenac sodium, according to a recent study. The clinical trial followed 45 subjects, randomized to three groups, for 8 weeks. Group one received diclofenac sodium, 50 mg, twice daily; group two received 500 mg BCM-95 high absorption curcumin twice daily; and group three received both diclofenac sodium and BCM-95 curcumin. In the BCM-95 curcumin groups, there were no dropouts due to adverse effects, but in the diclofenac sodium group, 14% withdrew due to adverse effects.
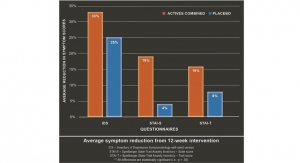
In the Disease Activity Score (DAS)28 assessment, which includes analysis of joint swelling and pain, BCM-95 curcumin had the highest impact for reducing disease symptoms, followed by BCM-95 curcumin with diclofenac sodium, with the diclofenac sodium-alone group third. However, though the numbers were higher in the BCM-95 group, none of the differences between the drug and curcumin group were statistically significant in terms of effectiveness.
The BCM-95 group showed improvement over others in reducing C-reactive protein (CRP) a measure of chronic inflammation, and antistreptococcal antibodies (ASO) titers, which are associated with severity of RA activity.
In the Disease Activity Score (DAS)28 assessment, which includes analysis of joint swelling and pain, BCM-95 curcumin had the highest impact for reducing disease symptoms, followed by BCM-95 curcumin with diclofenac sodium, with the diclofenac sodium-alone group third. However, though the numbers were higher in the BCM-95 group, none of the differences between the drug and curcumin group were statistically significant in terms of effectiveness.
The BCM-95 group showed improvement over others in reducing C-reactive protein (CRP) a measure of chronic inflammation, and antistreptococcal antibodies (ASO) titers, which are associated with severity of RA activity.