04.01.05
Indication: Depression
Source: BMJ, February 11, 2005 (Epub ahead of print).
Research: A randomized, double-blind, reference-controlled trial was conducted on 251 patients ages 18 to 70 during the years 2000 through 2003 to determine the effects of SJW versus paroxetine, a well known antidepressant pharmaceutical drug marketed under the brand name Paxil by GlaxoSmithKline. (A reference-controlled trial is different than a placebo-controlled trial where some of the patients take a placebo—a dose with no active ingredient—as a control to test the effect of the herb or drug being studied. In a reference-controlled trial, the substance being tested is compared directly to a known active material, in this case, the antidepressant paroxetine.) In the study, subjects were randomized to receive either 900 mg/day of WS 5570 (SJW) or 20 mg/day of paroxetine. If a patient did not experience a measurable response after two weeks, the dose was doubled.
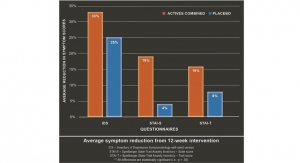
Results: Investigators found that patients in the SJW group experienced significantly better improvements as measured by the Hamilton Depression Rating Scale (HAMD), an internationally accepted severity scale for depression. In addition, SJW produced significantly fewer adverse effects than the drug. In fact, the paroxetine group 269 adverse effects compared to only 172 for those on SJW. Researchers concluded that SJW was as effective as a paroxetine in reducing symptoms of depression.
Source: BMJ, February 11, 2005 (Epub ahead of print).
Research: A randomized, double-blind, reference-controlled trial was conducted on 251 patients ages 18 to 70 during the years 2000 through 2003 to determine the effects of SJW versus paroxetine, a well known antidepressant pharmaceutical drug marketed under the brand name Paxil by GlaxoSmithKline. (A reference-controlled trial is different than a placebo-controlled trial where some of the patients take a placebo—a dose with no active ingredient—as a control to test the effect of the herb or drug being studied. In a reference-controlled trial, the substance being tested is compared directly to a known active material, in this case, the antidepressant paroxetine.) In the study, subjects were randomized to receive either 900 mg/day of WS 5570 (SJW) or 20 mg/day of paroxetine. If a patient did not experience a measurable response after two weeks, the dose was doubled.
Results: Investigators found that patients in the SJW group experienced significantly better improvements as measured by the Hamilton Depression Rating Scale (HAMD), an internationally accepted severity scale for depression. In addition, SJW produced significantly fewer adverse effects than the drug. In fact, the paroxetine group 269 adverse effects compared to only 172 for those on SJW. Researchers concluded that SJW was as effective as a paroxetine in reducing symptoms of depression.