Search Results for 'Quality'
-
Cardiovascular Health | Consumer Trends | Diabetes & Blood Sugar Management | Dietary Supplements | Healthcare Trends | Herbs & Botanicals | Proteins, Peptides, Amino Acids | Quality & Safety | Weight Management/Weight Loss

The Big Picture: Weight Wellness Trends
Consumers continue to tip the scales at a time when health hangs in the balance.By Sean Moloughney, Editor 09.08.20
-
Diabetes & Blood Sugar Management | Herbs & Botanicals | Immune Function | Quality & Safety | Research | Sports Nutrition | Testing

Botanical Research: Medicinal Mushrooms, Tart Cherry for Endurance, Nigella Sativa for Diabetes
AHPA compiles recent research on botanicals and plant compounds published in peer-reviewed journals.By Holly Johnson, PhD, American Herbal Products Association (AHPA) 09.08.20
-
Bone & Joint Health | Breaking News | Contract Manufacturing | Healthcare Trends | Industry & Market News | Mergers & Acquisitions | Proteins, Peptides, Amino Acids | Quality & Safety | Skin Health | World Markets

Gelatin Trade Associations Coalesce to Form Global Group
The Gelatin Representatives of the World (GROW) is comprised of gelatin associations from four different continents.09.02.20
-
Breaking News | Dietary Supplements | Herbs & Botanicals | Natural/Organic | Products | Quality & Safety | Regulations | Research | Supplier News | World Markets

PLT Health Solutions Receives Canadian Marketing License for Respiratory Support Ingredient
AlvioLife, a formulation of Boswellia serrata and quince fruit extracts, was shown by clinical work to support overall respiratory health.08.31.20
-
Breaking News | Herbs & Botanicals | Industry & Market News | Infant & Children's Health | Natural/Organic | Quality & Safety | World Markets

Infant Death Linked to Goldenseal Contaminated with Bacteria
The company is voluntarily recalling its products after FDA lab analysis found microbial contamination in its samples.08.24.20
-
Breaking News | Medical Nutrition | Quality & Safety | Regulations | Regulatory News

Colloidal Silver Promoter Charged with Making Fraudulent COVID-19 Health Claims
According to the FDA, SilveryGuy was participating in the Amazon Associates program, and is not the first participant to allegedly market products in this way.08.19.20
-
Breaking News | Healthcare Trends | Herbs & Botanicals | Industry & Market News | Quality & Safety

ABC, AHPA Warn Against Home Remedies Containing Highly-Toxic Oleander
The herb contains chemicals that can cause death, and serious cardiovascular side effects.08.18.20
-
Breaking News | Dietary Supplements | Herbs & Botanicals | Industry & Market News | Products | Proteins, Peptides, Amino Acids | Quality & Safety | Testing | World Markets

NOW Raises Additional Concerns About Supplements Available on Amazon
Following reports of labeling and quality problems on CoQ10 and SAMe, NOW is raising the alarm on Phosphatidyl Serine products.08.12.20
-
Breaking News | Diabetes & Blood Sugar Management | Dietary Supplements | Medical Nutrition | Probiotics & Prebiotics | Products | Quality & Safety | Research | Research News | Testing

The First Medical Probiotic for Type 2 Diabetes Management
Pendulum's formulation contains novel strains of bacteria, and a one-of-a-kind prebiotic fiber.08.07.20
-
Breaking News | Consumer Trends | Herbs & Botanicals | Industry & Market News | Natural/Organic | Quality & Safety | Regulations | Research | Testing | World Markets

AHPA to Host Virtual Hemp-CBD Summit to Help Companies Navigate the Marketplace
Speakers will include members of the FTC, the USDA, Sen. Ron Wyden, and industry leaders.08.04.20
-
Breaking News | Contract Manufacturing | Dietary Supplements | Industry & Market News | Mergers & Acquisitions | Quality & Safety | Regulations | Testing | World Markets

FoodChain ID Acquires Nutraveris
The European company specializes in similar domains as FoodChain ID, such as supply chain compliance and claim verification services.08.04.20
-
Breaking News | Dietary Supplements | Quality & Safety | Regulations | Regulatory News | Sports Nutrition | Weight Management/Weight Loss
Potential MA, NY Bans on Weight Loss, Muscle Building Supplements Spark Industry Backlash
State bills proposed in Massachusetts and New York would be the first to place popular bulking and cutting ingredients under lock-and-key from minors if passed.07.31.20
-
Breaking News | Diabetes & Blood Sugar Management | Functional Foods & Beverages | Healthy Aging | Natural/Organic | Quality & Safety | Research | Research News | Sweeteners | Weight Management/Weight Loss

Americans Are Consuming Less Sugar and More Non-Nutritive Sweeteners
A survey examined the sweetener purchasing habits by sales volume between the years 2002 and 2018.07.29.20
-
Breaking News | Dietary Supplements | Quality & Safety | Regulations | Regulatory News | Testing | World Markets

FDA Publishes Blueprint for New Era of Food Safety
The policy initiative, which was announced in 2019, will leverage new technology to improve food safety practices.07.28.20
-
Breaking News | Certifications, Approvals & Patents | Consumer Trends | Natural/Organic | Quality & Safety | Supplier News | Testing | World Markets

Sabinsa Earns Non-GMO Project Verification for 11 Ingredients
Non-GMO ingredients have proven to be a lasting top concern among eco-conscious consumers.07.27.20
-
Breaking News | Industry & Market News | Quality & Safety | Regulations | World Markets

NPA Survey: Safe Reopening Procedures Will Cost $13,595 Annually On Average
The trade organization is pushing for a federal tax credit to help businesses absorb the costs of COVID-19 Health Precautions.07.23.20
-
Dietary Supplements | Healthcare Trends | Quality & Safety | Regulations | Research

Dietary Supplement Clinical Research in the Age of COVID-19: Challenges & Opportunities
The fundamental importance of ensuring studies are well-designed has been magnified by challenges brought about by COVID-19.07.23.20
-
Industry & Market News | Quality & Safety | Regulations | Regulatory News | Testing

NPA Requests FDA Take More Action Against Adulteration
The trade group requested particularly that FDA begin issuing import alerts for supplements and expand participation in the group’s quality assurance program.07.21.20
-
Dietary Supplements | Herbs & Botanicals | Natural/Organic | Quality & Safety | Regulations | World Markets

FairWild Offers Certifications Unique to Wild Plants
The NGO merges social and ecological models promoted by the governments of Germany and Switzerland.By Mike Montemarano, Associate Editor 07.15.20
-
Dietary Supplements | Healthcare Trends | Herbs & Botanicals | Quality & Safety | World Markets

Sustaining Long-Term Health of Herbal Products
Investments and commitments to sustainability are critical for companies, and the plants they rely on, to thrive in years to come.By Sean Moloughney, Editor 07.15.20
-
Dietary Supplements | Herbs & Botanicals | Quality & Safety | World Markets

Nurturing the Herb-and-Herbalist Connection
Sustainable Herbs Program showcases best practices for stewardship of plants and communities.By Mike Montemarano, Associate Editor 07.15.20
-
Dietary Supplements | Herbs & Botanicals | Quality & Safety | Regulations

Potential Pitfalls for Hemp & CBD Products Within Liability Insurance Policies
Companies need to account for risks and know what’s covered, but sometimes that’s not a cut-and-dry situation.By Chris Morey, Bolton & Company 07.15.20
-
Healthcare Trends | Herbs & Botanicals | Natural/Organic | Quality & Safety | Testing

Oso Vega Inc: Elevating Agricultural Communities
Brittany Ferguson, VP of quality control and corporate values, discusses the company’s commodities management platform and value-added services.By Sheldon Baker, Baker Dillon Group LLC 07.15.20
-
Breaking News | Contract Manufacturing | Quality & Safety | Supplier News | Testing

NP Nutra Reports Growth in Sales Supported by Quality, Testing Program
Following strong growth in sales over the first half of 2020, NP Nutra gave the credit to its Triple-T Verification program.07.14.20
-
Breaking News | Contract Manufacturing | Industry & Market News | Quality & Safety | Regulations | Regulatory News | Testing

FDA Intends to Resume On-Site Domestic Inspections
Beginning July 20, the FDA is expected to resume pre-announced, prioritized inspections depending on COVID-19 trajectory data and state policy.07.13.20
-
Breaking News | Consumer Trends | Contract Manufacturing | Delivery & Dosage Technologies | Dietary Supplements | Men's Health | Minerals | Products | Quality & Safety | Vitamins

Ritual Expands to Male Consumers with the Launch of New Multivitamin
The company continues to uphold its mission statement that less is more, and clean labels and transparency are musts.Online Exclusives By Mike Montemarano , Associate Editor 07.09.20
-
Breaking News | Contract Manufacturing | Cosmeceuticals / Nutricosmetics | Herbs & Botanicals | Industry & Market News | Quality & Safety | Testing | World Markets

BAPP Publishes English Lavender Oil Adulteration Bulletin
The new bulletin summarizes common adulteration practices associated with English lavender, and methods of detecting fraudulent ingredients.07.07.20
-
Breaking News | Dietary Supplements | Digestive Health | Industry & Market News | Probiotics & Prebiotics | Quality & Safety | World Markets

Groups Take Issue with ’60 Minutes’ Segment on Probiotics
Trade associations said that, while the program recognized the safety of probiotics, it dismissed a wealth of research supporting the benefits.Online Exclusives By Mike Montemarano, Associate Editor 06.30.20
-
Breaking News | Certifications, Approvals & Patents | Contract Manufacturing | Quality & Safety | Regulations | Supplier News | Testing

ChromaDex R&D Facility Earns ISO/IEC 17025:2017 Accreditation
This quality management certification represents full compliance with the most rigorous ISO quality standards for labs of this kind.06.18.20
-
Breaking News | Herbs & Botanicals | Quality & Safety | Regulations | Research | Research News

No THC Bioconversion Occurred in Oral CBD Supplementation Study
In a study on 120 participants, CBD supplementation did not cause a presence of THC in blood plasma, and no psychotomimetic effects of THC occurred.06.16.20
-
Breaking News | Industry & Market News | Quality & Safety

NOW Named Best & Brightest Company To Work For
The National Association for Business Resources selected the company among 5,000 other nominees.06.09.20
-
Breaking News | Contract Manufacturing | Dietary Supplements | Industry & Market News | Quality & Safety

NPA Makes Recommendations Amid COVID-19 Adulteration Concerns
The trade association pitched import alerts and a greater participation in the SSCI throughout the pandemic and its related shortages.06.02.20
-
Herbs & Botanicals | Quality & Safety | Testing

Detecting Adulteration: Huperzine A Case Study
Carbon-14 testing offers a reliable means to differentiate between natural-derived and petrochemical-derived sources.Written By Haley Gershon, Marketing Manager, Beta Analytic, Research By Gao Yu, Beta Analytic & Xin Ma, Nanjing Hup Chemical Co., Ltd. 06.02.20
-
Healthcare Trends | Herbs & Botanicals | Quality & Safety | Research | Testing

Botanical Research: Cannabis Quality Considerations & Nutritional Status for Immune Health
AHPA compiles recent research on botanicals and plant compounds published in peer-reviewed journals.By Holly Johnson, PhD, American Herbal Products Association (AHPA) 06.02.20
-
Breaking News | Dietary Supplements | Functional Foods & Beverages | Industry & Market News | Quality & Safety | Regulations | Regulatory News | World Markets

FDA Temporarily Loosens Formulation, Vending Machine Regulations
It appears that the new policy will last throughout the COVID-19 public health emergency.05.28.20
-
Breaking News | Dietary Supplements | Herbs & Botanicals | Industry & Market News | Medical Nutrition | Natural/Organic | Quality & Safety | Regulations | Regulatory News | Research | World Markets

Hemp Industry Experts Call for Unity in Turbulent Years to Come
Murky regulatory status, competition from synthetic analogues, a global pandemic, and politics will present challenges to the hemp industry.Online Exclusives By Mike Montemarano, Associate Editor 05.26.20
-
Breaking News | Contract Manufacturing | Dietary Supplements | Industry & Market News | Quality & Safety | Regulations | Regulatory News | World Markets

Lief Labs Launches Complimentary GMP Starter Kit
The California-based supplement manufacturer published this free manual to provide the industry with a foundational guide to compliance with FDA standards.05.19.20
-
Breaking News | Healthcare Trends | Products | Quality & Safety

Gaia Herbs Donates Hand Sanitizer to Local Frontline Workers
Company has increased production and shifted its alcohol-based operations to manufacture CDC-approved hand sanitizer.05.19.20
-
Breaking News | Contract Manufacturing | Industry & Market News | Quality & Safety | Regulations | Regulatory News | Supplier News | Testing

Alkemist Labs Warns Industry about Doctored Lab Reports
The company is sounding the alarm after allegedly encountering multiple doctored versions of lab reports by product marketers and ingredients suppliers.05.13.20
-
Breaking News | Dietary Supplements | Green Foods | Herbs & Botanicals | Quality & Safety | Research | Research News | Supplier News | Testing | World Markets

Euromed Adopts Fig through ABC’s Adopt-an-Herb Program
The botanical supplement company will assist in the dissemination of industry know-how for all things pertinent to figs.05.12.20
-
Breaking News | Dietary Supplements | Industry & Market News | Products | Quality & Safety | Regulations | Testing
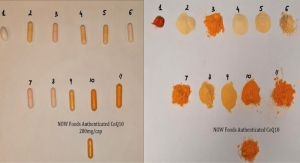
NOW Finds Quality Issues in Amazon-Purchased Products
An internal test of CoQ10 and SAMe supplements found significant issues with a number of products, including potency far below label claims.05.11.20
-
Breaking News | Cognitive Function | Cosmeceuticals / Nutricosmetics | Dietary Supplements | Healthy Aging | Immune Function | Medical Nutrition | Quality & Safety | Research | Research News | Supplier News | Vitamins
ChromaDex Highlights The Latest Research on Niagen
The patented nicotinamide riboside ingredient is being evaluated for applications including obesity, neurological protection, and immune response to COVID-19.05.08.20
-
Breaking News | Certifications, Approvals & Patents | Contract Manufacturing | Quality & Safety | Supplier News | Testing | World Markets

Wiley Companies Earns ISO 9001:2015 Certification
This certification represents approval of the company's quality management system from the International Organization for Standardization.05.07.20
-
Breaking News | Contract Manufacturing | Herbs & Botanicals | Industry & Market News | Quality & Safety | Research | World Markets

American Herbal Pharmacopoeia Collaborating with Tehran University on Chamomile Literature
The compendium will establish a set of industry standards to promote quality and integrity in all chamomile-based botanical products.05.06.20
-
Breaking News | Herbs & Botanicals | Industry & Market News | Quality & Safety

Botanical Safety Consortium to Meet Virtually May 29
Objectives include recruiting new members and soliciting input into current scientific challenges and opportunities related to botanical ingredient safety.05.05.20
-
Breaking News | Cardiovascular Health | Certifications, Approvals & Patents | Inflammation | Quality & Safety | Testing | World Markets

ThymoQuin Achieves Self-Affirmed GRAS Status from TriNutra
Tri-Nutra's product is the first black seed oil standardized to a high thymoquinone content.05.04.20
-
Breaking News | Consumer Trends | Flavors & Colors | Functional Foods & Beverages | Mergers & Acquisitions | Natural/Organic | Quality & Safety | Supplier News | World Markets

Kerry Acquires IsoAge Technologies and Biosecur Labs
The Ireland-based ingredients supplier sought these companies out to formulate clean-label solutions for food preservation, in lieu of artificial ingredients.05.04.20
-
Consumer Trends | Dietary Supplements | Healthcare Trends | Healthy Aging | Immune Function | Mood Health & Sleep

Pandemic Shifts Priorities for Health & Wellness
The crisis may have lasting implications for how consumers manage their well-being.By Sean Moloughney, Editor 05.01.20
-
Consumer Trends | Healthcare Trends | Herbs & Botanicals | Quality & Safety

Healist: A ‘Benefits-Based’ Brand Delivering CBD Formulations
Michael Bryce discusses the company’s core values of quality, transparency, and sustainability.By Sheldon Baker, Baker Dillon Group LLC 05.01.20
-
Breaking News | Contract Manufacturing | Herbs & Botanicals | Industry & Market News | Quality & Safety | Regulations | Research | Testing | World Markets

AOAC’s Method for Hemp Cannabinoid Analysis Approved
Hemp can now be evaluated with an officially-recognized method for THC on a dry-weight basis, as per the requirements of the 2018 Farm Bill.04.29.20












